Photoacoustic Imaging Illuminates Elusive Biological Processes
By Geoffrey P. Luke and Stanislav Y. Emelianov
The emerging field of photoacousitc imaging offers high resolution visualization of body tissues, while being non-invasive and not involving ionizing radiation.
Introduction
The ability to noninvasively detect functional or molecular changes which indicate the presence of disease is a major hurdle in biomedical research and clinical practice. To address this need, molecular imaging research has experienced a dramatic rise in activity over the course of the last decade. One such imaging technique – photoacoustic imaging – has garnered widespread interest because it can provide useful functional information or, with the advent of nanometer scale contrast agents, detailed information about the molecular expressions in tissue.[1][2]
Photoacoustic imaging relies on the photoacoustic effect first described by Alexander Graham Bell in 1880. Bell discovered that sounds were emitted when substances were exposed to “a rapidly-interrupted beam of sunlight.”[3] Indeed, modern photoacoustic imaging relies on a pulsed laser to generate broadband acoustic waves deep in tissue which are detected by an ultrasound transducer at the tissue’s surface. The acoustic waves are generated when optical absorbers, such as hemoglobin, melanin, or contrast agents, absorb the optical energy, resulting in a localized transient temperature rise. The increase in temperature causes thermoelastic expansion of the tissue and the emission of an ultrasound wave.
The unique set of benefits associated with photoacoustic imaging makes it an excellent alternative to other biomedical imaging modalities.[4] First, it does not use ionizing radiation, which makes it a safe alternative to radiographic or nuclear imaging methods. Second, the spatial resolution, which is determined primarily by the ultrasound transducer, is excellent at clinically relevant depths – in contrast, purely optical imaging methods suffer from poor resolution at depths greater than several millimeters. Third, photoacoustic imaging provides high contrast images of vasculature or contrast agents. Finally, the systems are typically portable and images can be acquired in real-time. Thus, photoacoustic imaging promises to fill a void in current clinical biomedical imaging.
Imaging Tissue Components as Indicators of Disease
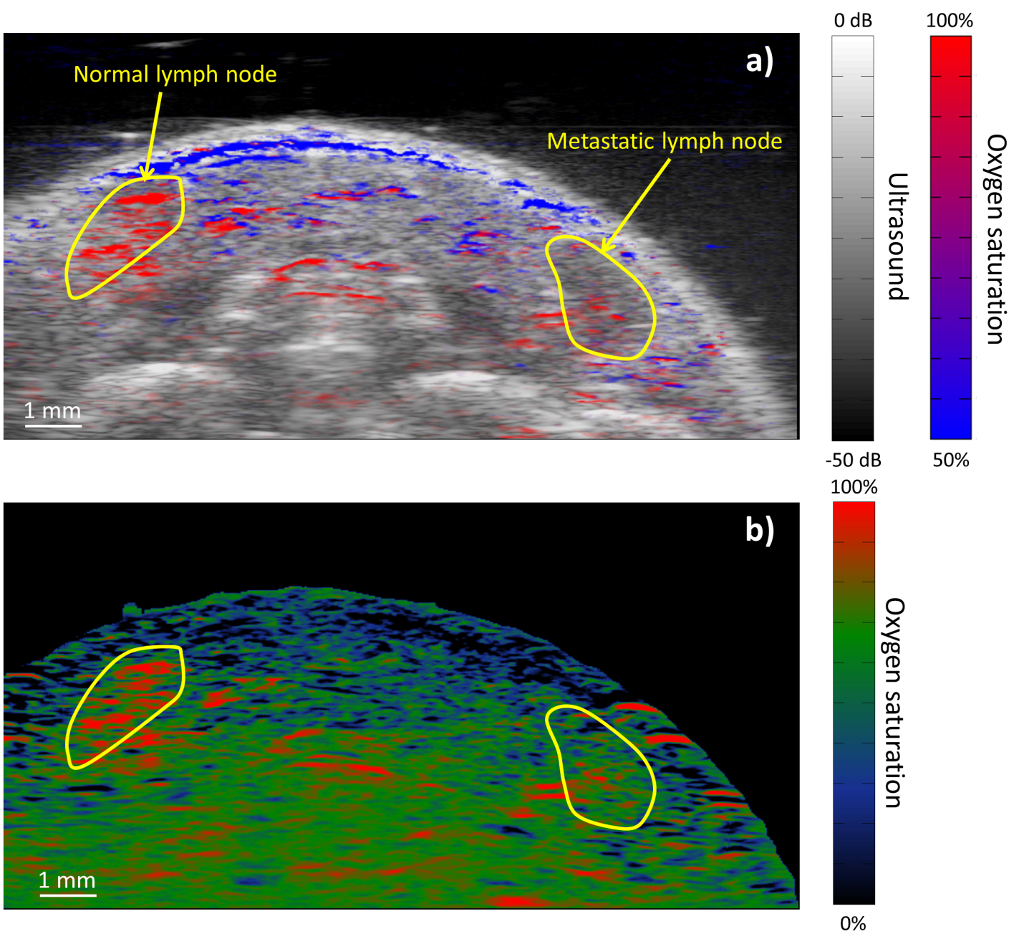
The unique contrast mechanism in photoacoustic imaging – optical absorption – allows for the detection and characterization of tissue types that is not available with any other imaging modality. The main source of signal in a typical photoacoustic image is generated by hemoglobin. Thus, high resolution images of the vasculature can be obtained. This will allow physicians to identify abnormal changes in vasculature (e.g., the disorganized angiogenesis associated with tumor development). Furthermore, if multiple photoacoustic images are acquired using separate excitation wavelengths spanning the red to near infrared, then spectroscopic methods can be used to differentiate between optical absorbers in tissue. For example, the absorption spectrum of blood, which varies as a function of its oxygen content, can be analyzed to separate veins from arteries (Fig. 1a) and identify regions of hypoxia (Fig. 1b) – an important indicator of malignancy in cancer.
Photoacoustic imaging also has the potential to aid in the diagnosis of cardiovascular disease. Lipid pools, a key component of vulnerable plaques, can be detected by tuning the imaging laser to 1720 nm, where lipid absorption exceeds all other endogenous absorbers. Initial in vivo experiments in a rabbit model of atherosclerosis indicate that photoacoustic imaging could dramatically improve the way in which cardiovascular disease is diagnosed and treated.[5]
Contrast Agents
The versatility of photoacoustic imaging can be extended with the introduction of contrast agents. Strongly absorbing materials, including dyes and nanoparticles, generate strong photoacoustic signals. Nanoparticles constructed from noble metals (e.g., gold or silver) make particularly good contrast agents because their optical absorption is orders of magnitude greater than dyes – a result of the plasmon resonance effect – and their surface can chemically modified to enhance delivery and target specific biomarkers which are indicative of disease.[2]
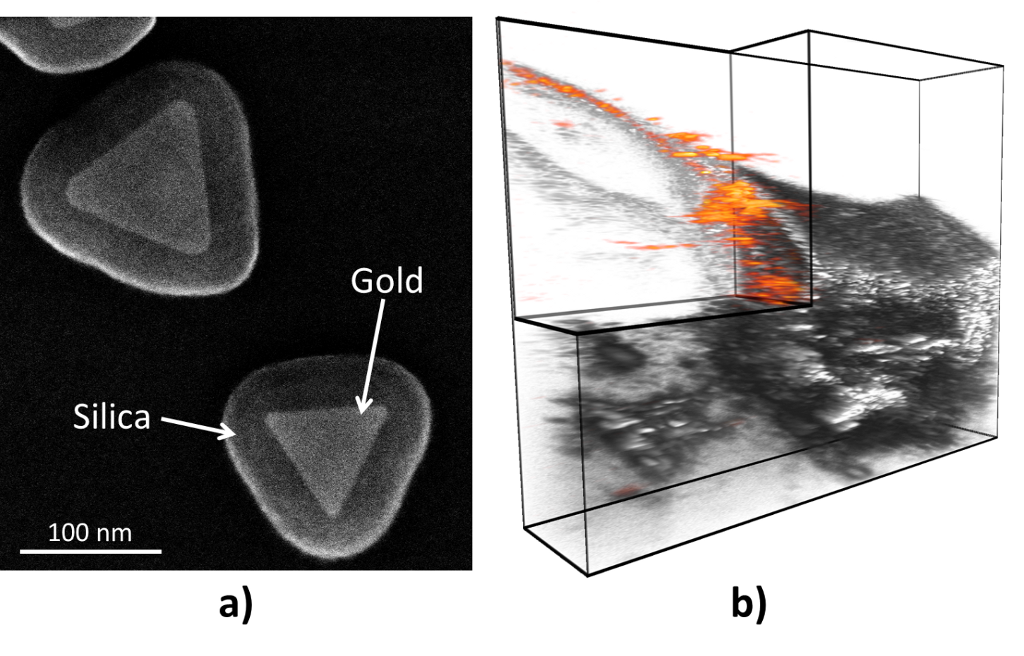
By changing the size and shape of the nanoparticle, the optical properties can be controlled. In this manner, the optical absorption can be tuned to 1064 nm, where light penetration in tissue is at a maximum and inexpensive Nd:YAG lasers can be used for imaging. Indeed, when silica-coated gold nanoplates (Fig. 2a) are used to map lymph nodes in mice, extremely high photoacoustic contrast can be achieved (Fig. 2b). In fact, photoacoustic signal originating from endogenous absorbers was not detectable in this experiment.[6]
Recently, investigations into the physics of photoacoustic signal formation have led to insights into optimizing nanoparticle contrast agents. Although the nanoparticle is responsible for the absorption of the light, the bulk of the photoacoustic wave is generated in the medium surrounding the nanoparticle.[7] Thus, the transfer of heat from the nanoparticle to the surrounding tissue becomes a critical factor. Indeed, by coating gold nanoparticles with a thin (c.a. 20 nm) layer of silica, the interfacial heat resistance between the nanoparticle and the surrounding medium can be reduced, the photothermal stability of the nanoparticle is improved, and the overall photoacoustic signal is amplified.[8]
The Clinical Translation
So far, photoacoustic imaging has been primarily restricted to the preclinical domain. Three companies – iThera Medical, Endra, and VisualSonics – currently offer preclinical commercial systems for small animal imaging. As with any new medical technology, clinical acceptance is slow and proceeds in an application-dependent manner. Initial forays into clinical imaging have focused on evaluating the role of photoacoustic imaging in breast cancer screening.[9] By imaging elevated blood, photoacoustic imaging has been able to identify suspicious lesions in breast cancer patients.
It should be emphasized that nanoparticle contrast agents face a longer regulatory approval and, therefore, clinical acceptance than label-free photoacoustic imaging. Although noble metal nanoparticles do not exhibit acute toxicity, gold particles larger than 5 nm tend to accumulate in the spleen and liver indefinitely.[10] Therefore, more studies need to be performed to establish the safety of such contrast agents. However, given the power of these contrast agents, they may one day play an important role in the clinic.
With the frontier of medicine focused on treating diseases on molecular and functional levels, there is an increasing need for imaging techniques which can accurately detect these changes in patients. Photoacoustic imaging promises to fill this role for a wide variety of pre-clinical and clinical applications because of the essential information it provides. We envision that it will be an integral tool in the future of personalized medicine.
For Further Reading
1. S. Mallidi, G. P. Luke, S. Emelianov, “Photoacoustic imaging in cancer detection, diagnosis, and treatment guidance”, Trends in Biotechnology, 2011;29(5):213-21.
2. G. P. Luke, D. Yeager, S. Y. Emelianov, “Biomedical applications of photoacoustic imaging with exogenous contrast agents”, Ann Biomed Eng. ,2012;40(2):422-37.
3. A. G. Bell, “Upon the production of sound by radiant energy”, Philosophical Magazine Series 5, 1881;11(71):510-28.
4. S. Y. Emelianov, P-C Li, M. O’Donnell, “Photoacoustics for molecular imaging and therapy”, Physics Today, 2008:34-9.
5. B. Wang, A. Karpiouk, D. Yeager, J. Amirian, S. Litovsky, R. Smalling, et al, “In vivo intravascular ultrasound-guided photoacoustic imaging of lipid in plaques using an animal model of atherosclerosis”. Ultrasound in medicine & biology, 2012.
6. G. P. Luke, A. Bashyam, K. A. Homan, S. Makhija, Y. S. Chen, S. Y. Emelianov, “Silica-coated gold nanoplates as stable photoacoustic contrast agents for sentinel lymph node imaging”, Nanotechnology, 2013 (Accepted).
7. Y-S Chen, W. Frey, S. Aglyamov, S. Emelianov, “Environment-dependent generation of photoacoustic waves from plasmonic nanoparticles”, Small, 2012;8(1):47-52.
8. Y-S Chen, W. Frey, S. Kim, P. Kruizinga, K. Homa, S. Emelianov, “Silica-coated gold nanorods as photoacoustic signal nanoamplifiers”, Nano Lett., 2011;11(2):348-54.
9. M. Heijblom, D. Piras, W. Xia, J. van Hespen, J. Klaase, F. Van den Engh, et al, “Visualizing breast cancer using the twente photoacoustic mammoscope: What do we learn from twelve new patient measurements?”, Optics express, 2012;20(11):11582-97.
10. N. Khlebtsov, L. Dykman, “Biodistribution and toxicity of engineered gold nanoparticles: A review of in vitro and in vivo studies”, Chemical Society Reviews, 2011;40(3):1647-71.
Contributors

Geoffrey Luke is a graduate student in the Department of Electrical Engineering at The University of Texas at Austin. He received a B.S. in Computer Engineering and Mathematics and a M.S. in Electrical Engineering from the University of Wyoming, where he developed a sensor based on the visual system of the common housefly. His current research in the Ultrasound Imaging and Therapeutics Research Laboratory is focused on early cancer detection and characterization using molecular spectroscopic photoacoustic imaging. Read more

Stanislav Y. Emelianov (B.S. and Ph.D. from The Moscow State University, Russia) is currently a Professor and an Associate Chair for Research in the Department of Biomedical Engineering at The University of Texas at Austin where he directs the Ultrasound Imaging and Therapeutics Research Laboratory. In addition, Dr. Emelianov is an Adjunct Professor of Imaging Physics at The University of Texas M.D. Anderson Cancer Center in Houston. Dr. Emelianov’s research interests are in the areas of medical imaging for diagnostic and therapeutic applications, bionanotechnology, photoacoustic imaging, ultrasound imaging, elasticity imaging, cellular/molecular imaging, and functional imaging. Read more







 Dr. William J. Heetderks is the Director of Extramural Science Programs at the National Institute of Biomedical Imaging and Bioengineering (NIBIB), NIH. The extramural program supports approximately 800 research and training grants at universities and research centers throughout the United States in fields of bioengineering and biomedical imaging.
Dr. William J. Heetderks is the Director of Extramural Science Programs at the National Institute of Biomedical Imaging and Bioengineering (NIBIB), NIH. The extramural program supports approximately 800 research and training grants at universities and research centers throughout the United States in fields of bioengineering and biomedical imaging.  Geoffrey Luke is a graduate student in the Department of Electrical Engineering at The University of Texas at Austin. He received a B.S. in Computer Engineering and Mathematics and a M.S. in Electrical Engineering from the University of Wyoming, where he developed a sensor based on the visual system of the common housefly.
Geoffrey Luke is a graduate student in the Department of Electrical Engineering at The University of Texas at Austin. He received a B.S. in Computer Engineering and Mathematics and a M.S. in Electrical Engineering from the University of Wyoming, where he developed a sensor based on the visual system of the common housefly.  Stanislav Y. Emelianov (B.S. and Ph.D. from The Moscow State University, Russia) is currently a Professor and an Associate Chair for Research in the Department of Biomedical Engineering at The University of Texas at Austin where he directs the Ultrasound Imaging and Therapeutics Research Laboratory. In addition, Dr. Emelianov is an Adjunct Professor of Imaging Physics at The University of Texas M.D. Anderson Cancer Center in Houston.
Stanislav Y. Emelianov (B.S. and Ph.D. from The Moscow State University, Russia) is currently a Professor and an Associate Chair for Research in the Department of Biomedical Engineering at The University of Texas at Austin where he directs the Ultrasound Imaging and Therapeutics Research Laboratory. In addition, Dr. Emelianov is an Adjunct Professor of Imaging Physics at The University of Texas M.D. Anderson Cancer Center in Houston.  Mathukumalli Vidyasagar is the Founding Head of the Bioengineering Department, University of Texas at Dallas. He is a Fellow of the Royal Society, UK.
Mathukumalli Vidyasagar is the Founding Head of the Bioengineering Department, University of Texas at Dallas. He is a Fellow of the Royal Society, UK.  Ayache Bouakaz graduated from the University of Sétif, Algeria, from the Department of Electrical Engineering. He obtained his Ph.D. degree in 1996 from the Department of Electrical Engineering at the Institut National des Sciences Appliquées de Lyon (INSA Lyon), France.
Ayache Bouakaz graduated from the University of Sétif, Algeria, from the Department of Electrical Engineering. He obtained his Ph.D. degree in 1996 from the Department of Electrical Engineering at the Institut National des Sciences Appliquées de Lyon (INSA Lyon), France.  Jean-Michel Escoffre earned his M.Sc. degrees in Vectorology, Gene Therapy, Vaccinology and Cell Biophysics in 2005 and 2006 respectively, at the University Paul Sabatier (Toulouse, France). He obtained his Ph.D. degree in Cell Biophysics in 2010 at the Institute of Pharmacology and Structural Biology (CNRS, UMR 5089) of the University Paul Sabatier.
Jean-Michel Escoffre earned his M.Sc. degrees in Vectorology, Gene Therapy, Vaccinology and Cell Biophysics in 2005 and 2006 respectively, at the University Paul Sabatier (Toulouse, France). He obtained his Ph.D. degree in Cell Biophysics in 2010 at the Institute of Pharmacology and Structural Biology (CNRS, UMR 5089) of the University Paul Sabatier.