Testing the Pattern of AKT Activation by Variational Parameter Estimation
By Daniel Kaschek, Frauke Henjes, Max Hasmann, Ulrike Korf, and Jens Timmer
Published November 10, 2016.
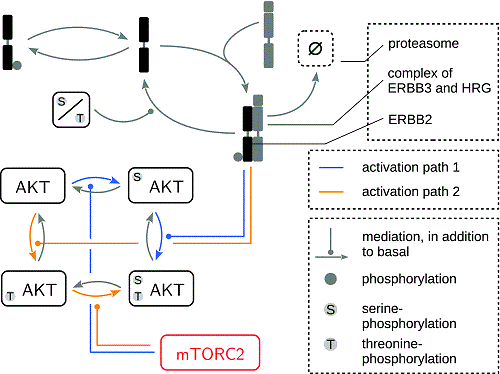
Dynamic modeling has become one of the pillars of understanding complex biological systems from a mechanistic point of view. In particular, ordinary differential equations are frequently used to model the dynamics of the interacting states, e.g., molecular species in cell signaling pathways. The equations typically contain many unknown parameters, such as reaction rates and initial conditions, but also time-dependent parameters, i.e., input functions driving the system. Both are a priori unknown and need to be estimated from experimental, time-resolved data. Here, we discuss an application of indirect optimal control methods for input estimation and parameter estimation in the mammalian target of rapamycin (mTOR) signaling. Whereas the direct identification and quantification of different active mTOR complexes, e.g., mTOR complex 2 (mTORC2), is only possible by highly challenging experiments, the mathematical framework allows to reconstruct its dynamics by solving an appropriate Euler–Lagrange equation based on Pontryagin’s maximum principle. The inherently large search space underlying this approach allows to test specific biological hypotheses about the activation of protein kinase B (AKT) by mTORC2 and to reject an alternative model with high statistical power. Hereby, we identify a minimal model that has AKT threonine phosphorylation as a prerequisite for serine phosphorylation by mTORC2. Based on this model, the activation of mTORC2 is predicted to be inhibited by drugs, targeting the receptors of the ERBB receptor family.








 Interim Editor-in-Chief
Interim Editor-in-Chief  Senior Editor for Personalized Medicine
Senior Editor for Personalized Medicine  Senior Editor for Pharmaceutical Engineering
Senior Editor for Pharmaceutical Engineering  Senior Editor for Synthetic Biology
Senior Editor for Synthetic Biology  Senior Editor for Systems Biology
Senior Editor for Systems Biology