Wireless Intracranial Pressure Monitoring in Blast-induced Traumatic Brain Injury in Rats
By Usmah Kawoos*, Xu Meng, Arye Rosen, and Mikulas Chavko*
A key parameter in monitoring brain injury is the measurement of intra-cranial pressure. This article describes a small, long-term implantable sensor developed for chronic monitoring in small animals.
The prevalence of traumatic brain injury (TBI) has been on rise in the past decade. Reportedly, a majority of TBI cases during the recent conflicts are due to explosive blasts [1]. The signature blast overpressure wave is being produced in the laboratory settings by utilizing a shock tube [2]. The intracranial pressure (ICP) being one of the key parameters for the monitoring of brain injury, has been measured in-vivo during exposure to the blast overpressure (BOP) by fiber optic pressure transducers [3]. These pressure probes are adequate for the recording of ICP in acute situations, but they cannot be relied on for chronic conditions. The pressure probes are limited in the scope of their application towards chronic measurements due to their fragility, wired connectivity, and the need to immobilize the subjects. This article describes an implantable device for a chronic in-vivo measurement of the ICP.
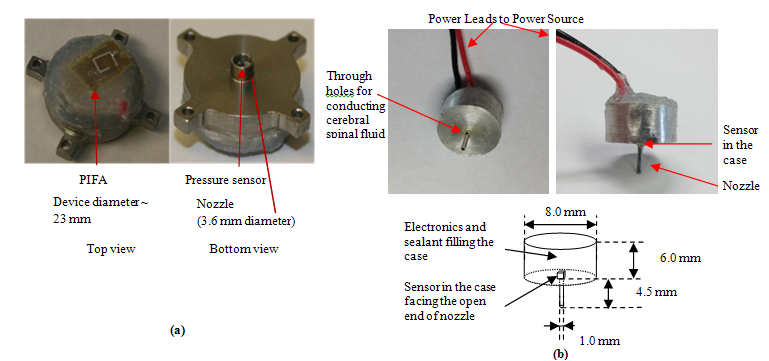
Figure 1: Description of the ICP monitoring devices. (a) photo of the device for large animals with integrated power supply, (b)photo and sketch of the device for small animals with external power supply.
A telemetric device operating at 2.4 GHz ISM band was described in [3] which is a completely implantable ICP monitoring device designed for large animal models. Rodents are a common laboratory animal model for conducting blast-induced TBI studies; consequently, a large body of data on the neuropathology of TBI has been published. The packaging of the device described before [3] was redesigned to be implantable in rats. Fig.1a., shows the picture of the device described by Kawoos et al., [3] which was implanted in canine models compared with the modified device which was implanted in rats (Fig.1b). The larger device has an embedded power supply and a power management system. On the other hand, the modified device does not contain any power source. Due to the small size of a rat’s head, the device was packaged only with the components required to sense (capacitive MEMS pressure sensor, Murata, Vantaa, Finland), process (Schmitt Trigger and a voltage controlled oscillator) and transmit (PIFA-antenna) the pressure in the intra-ventricular space. A pair of power terminals emerging from the device package (cylinder (8 mm diameter and 6 mm height) with an open nozzle (1mm diameter and 4.5 mm height), at the base of the cylinder) were connected to the external power source which was connected only at the time of recording the ICP. Fig.2., shows a packaged device, and a sketch of the device placement on the rat`s skull. The pressure signal was captured on a digital oscilloscope (DPO 7254, Tektronix, OR).
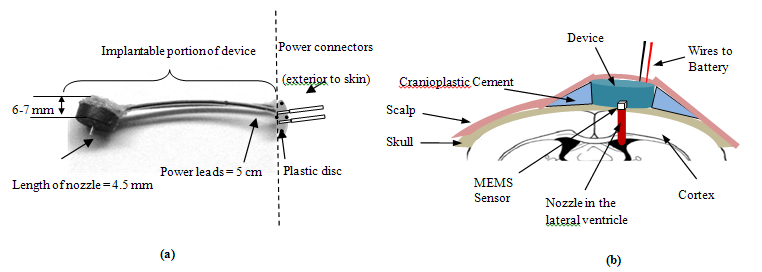
Figure 2: The details of the modified ICP monitoring device. (a) packaged device, (b) implantation details of the device with its nozzle placed in the lateral ventricle.
The devices were tested and calibrated in-vitro and the preliminary in-vivo study in rats ^(for a period of 7 days) showed favorable performances. The proper implantation of the device and its response to the variations in ICP were verified by inducing a transient increase in the ICP. The ICP measured in control: non-injured animals, was within the normal physiological range of the rats. The ICP showed a marked increase in rats suffering from TBI due to blast exposure. The trends in ICP variation were also related to the dose of blast exposure (i.e., intensity and number of blasts). The devices maintained good performance after being exposed to the BOP and remained biocompatible and integral within a biological environment (Kawoos et al, not published yet). In summary, a small device, operating at microwave frequency, was designed and developed for chronic monitoring of the ICP in small animal models. This device was utilized to record the trends in ICP, over the duration of one week, in blast-induced TBI models of injury. The applicability of this device can be extended to other forms of neurological disorders like hydrocephalus, non-blast related TBI, Reye’s syndrome etc with potentially less invasive surgical procedure than existing devices for continuous ICP recording. This device can also be utilized in determining the efficiency of treatments in treating the cause of abnormal ICP.
^ The experiments reported herein were conducted in compliance with the Animal Welfare Act and in accordance with the principles set forth in the “Guide for the Care and Use of Laboratory Animals,” Institute of Laboratory Animals Resources, National Research Council, National Academy Press, 1996.
* I am a military service member (or employee of the U.S. Government). This work was prepared as part of my official duties. Title 17 U.S.C. §105 provides that ‘Copyright protection under this title is not available for any work of the United States Government.’ Title 17 U.S.C. §101 defines a U.S. Government work as a work prepared by a military service member or employee of the U.S. Government as part of that person’s official duties.
For Further Reading
1. J. Magnuson, F. Leonessa, and G. S. Ling, “Neuropathology of explosive blast traumatic brain injury,” Curr Neurol Neurosci Rep, vol. 12, pp. 570-9, Oct 2012.
2. M. Chavko, W. A. Koller, W. K. Prusaczyk, and R. M. McCarron, “Measurement of blast wave by a miniature fiber optic pressure transducer in the rat brain,” J Neurosci Methods, vol. 159, pp. 277-81, Jan 30 2007.
3. U. Kawoos, M. R. Tofighi, R. Warty, F. A. Kralick, and A. Rosen, “Trans-Scalp Evaluation of an Intracranial Pressure Implant at 2.4 GHz,” Microwave Theory and Techniques, IEEE Transactions on, vol. 56, pp. 2356-2365, 2008.
Contributors
 Usmah Kawoos (S ’05-M ’09) received the Ph.D degree in biomedical engineering from Drexel University, Philadelphia, PA. Dr. Kawoos is currently working as a post-doctoral fellow at the Naval Medical Research Center, Silver Spring, MD. Her research interest are the application of microwaves in medicine and the biocompatibility of medical implants. Read more
Usmah Kawoos (S ’05-M ’09) received the Ph.D degree in biomedical engineering from Drexel University, Philadelphia, PA. Dr. Kawoos is currently working as a post-doctoral fellow at the Naval Medical Research Center, Silver Spring, MD. Her research interest are the application of microwaves in medicine and the biocompatibility of medical implants. Read more
 Xu Meng (S ’08-M ’13) received the Ph.D. degree in biomedical engineering in 2013 from Drexel University, Philadelphia, PA. His research interests are the clinical application of microwave and RF in medical implants and telemedicine. Read more
Xu Meng (S ’08-M ’13) received the Ph.D. degree in biomedical engineering in 2013 from Drexel University, Philadelphia, PA. His research interests are the clinical application of microwave and RF in medical implants and telemedicine. Read more
 Arye Rosen (M ’77-SM ’80-F ’92) received the Ph.D. degree in electrical engineering from Drexel University, Philadelphia, PA. Dr. Rosen currently holds an appointment as Academy Professor of Biomedical and Electrical Engineering in the School of Biomedical Engineering, Science and Health Systems and Associate Vice Provost for Strategic Initiatives at Drexel University, Philadelphia, PA. He also holds the title of Associate in Medicine at Jefferson Medical College, where he has been engaged in research in the Division of Cardiology since 1970. Read more
Arye Rosen (M ’77-SM ’80-F ’92) received the Ph.D. degree in electrical engineering from Drexel University, Philadelphia, PA. Dr. Rosen currently holds an appointment as Academy Professor of Biomedical and Electrical Engineering in the School of Biomedical Engineering, Science and Health Systems and Associate Vice Provost for Strategic Initiatives at Drexel University, Philadelphia, PA. He also holds the title of Associate in Medicine at Jefferson Medical College, where he has been engaged in research in the Division of Cardiology since 1970. Read more
 Mikulas Chavko received the PhD degree in Biochemistry from University of Veterinary Medicine in Kosice, Czechoslovakia. Currently, he serves as a Principal Investigator and Head of Blast Research in the Neurotrauma Department at the Naval Medical Research Institute. Read more
Mikulas Chavko received the PhD degree in Biochemistry from University of Veterinary Medicine in Kosice, Czechoslovakia. Currently, he serves as a Principal Investigator and Head of Blast Research in the Neurotrauma Department at the Naval Medical Research Institute. Read more







 Dr. Stephen Quake, D.Phil., is a Professor of Bioengineering and of Applied Physics at Stanford University. He pioneered the development of Microfluidic Large Scale Integration (LSI), demonstrating the first integrated microfluidic devices with thousands of mechanical valves.
Dr. Stephen Quake, D.Phil., is a Professor of Bioengineering and of Applied Physics at Stanford University. He pioneered the development of Microfluidic Large Scale Integration (LSI), demonstrating the first integrated microfluidic devices with thousands of mechanical valves.  H. Chitsaz is assistant professor of Computer Science at Wayne State University, Detroit, MI. He received his Ph.D. in Computer Science and M.S. in Mathematics from the University of Illinois at Urbana-Champaign.
H. Chitsaz is assistant professor of Computer Science at Wayne State University, Detroit, MI. He received his Ph.D. in Computer Science and M.S. in Mathematics from the University of Illinois at Urbana-Champaign.  Mathukumalli Vidyasagar is the Founding Head of the Bioengineering Department, University of Texas at Dallas. He is a Fellow of the Royal Society, UK.
Mathukumalli Vidyasagar is the Founding Head of the Bioengineering Department, University of Texas at Dallas. He is a Fellow of the Royal Society, UK.  Usmah Kawoos (S '05-M '09) received the Ph.D degree in biomedical engineering from Drexel University, Philadelphia, PA. Dr. Kawoos is currently working as a post-doctoral fellow at the Naval Medical Research Center, Silver Spring, MD.
Usmah Kawoos (S '05-M '09) received the Ph.D degree in biomedical engineering from Drexel University, Philadelphia, PA. Dr. Kawoos is currently working as a post-doctoral fellow at the Naval Medical Research Center, Silver Spring, MD.  Xu Meng (S '08-M '13) received the Ph.D. degree in biomedical engineering in 2013 from Drexel University, Philadelphia, PA. His research interests are the clinical application of microwave and RF in medical implants and telemedicine.
Xu Meng (S '08-M '13) received the Ph.D. degree in biomedical engineering in 2013 from Drexel University, Philadelphia, PA. His research interests are the clinical application of microwave and RF in medical implants and telemedicine.  Arye Rosen (M '77-SM '80-F '92) received the Ph.D. degree in electrical engineering from Drexel University, Philadelphia, PA. Dr. Rosen currently holds an appointment as Academy Professor of Biomedical and Electrical Engineering in the School of Biomedical Engineering, Science and Health Systems and Associate Vice Provost for Strategic Initiatives at Drexel University, Philadelphia, PA.
Arye Rosen (M '77-SM '80-F '92) received the Ph.D. degree in electrical engineering from Drexel University, Philadelphia, PA. Dr. Rosen currently holds an appointment as Academy Professor of Biomedical and Electrical Engineering in the School of Biomedical Engineering, Science and Health Systems and Associate Vice Provost for Strategic Initiatives at Drexel University, Philadelphia, PA.  Mikulas Chavko received the PhD degree in Biochemistry from University of Veterinary Medicine in Kosice, Czechoslovakia. Currently, he serves as a Principal Investigator and Head of Blast Research in the Neurotrauma Department at the Naval Medical Research Institute.
Mikulas Chavko received the PhD degree in Biochemistry from University of Veterinary Medicine in Kosice, Czechoslovakia. Currently, he serves as a Principal Investigator and Head of Blast Research in the Neurotrauma Department at the Naval Medical Research Institute.