Photonic Crystal Enhanced Microscopy: A New Form of Microscopy for Quantitative Imaging of Cell-Surface Interactions
By Brian T. Cunningham
A new method allows researchers to view cellular growth and interactions, without requiring the use of stains or fixation!
Important biological processes such as cancer cell metastasis, wound healing, and cell death involve the interaction of cells with membranes and extracellular matrix material within tissue. Characterizing these processes in the laboratory traditionally involves fluorescent dyes, fluorescent proteins, histological stains, or fixation. Such approaches are either cytotoxic, or temporally constrained by the effects of fluorescence photobleaching. While existing forms of microscopy, such as phase contrast imaging, confocal microscopy, and total internal reflection fluorescence (TIRF) elucidate the mechanics and outcomes of cellular processes, the lack of long-term, time-course data collection poses a serious compromise to the study of natural cell behavior during processes that occur over extended time scales. To address these important needs, our group developed a new form of microscopy called Photonic Crystal Enhanced Microscopy (PCEM) [1]. In PCEM, instead of using an ordinary glass microscope slide as a surface for cell attachment, we fabricate a polymer-based photonic crystal (PC) that can be attached to an ordinary glass cover slip and prepared with a thin layer extracellular coating that mimics the surfaces that cells encounter in the body.
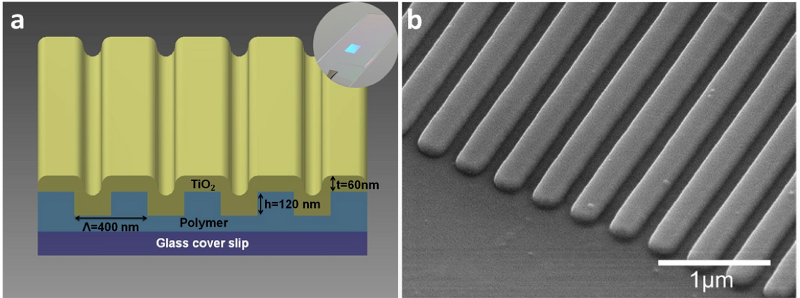
Figure 1:(a) Schematic diagram of the photonic crystal (PC) biosensor. A PC sensor is comprised of a replica molded polymer grating overcoated with a high refractive index thin film of TiO2. Inset: Photo of a PC fabricated upon a glass cover slip. (b) Scanning electron micrograph of the PC surface. Reproduced with permission from [1].
As shown in Figure 1, the PC is a periodically nanostructured surface that is designed to function as a wavelength tunable resonant reflectance filter. When illuminated with a broad band of wavelengths from a low intensity LED, the PC reflects only a narrow band of wavelengths near 632 nm. At the resonant wavelength, an electromagnetic standing wave is established that is confined near the PC surface, decaying exponentially as one moves away. When a cell attaches to the PC, the resonant wavelength shifts to a greater value, but only on the part of the PC surface where the cell is located, as the cell’s material components (lipid membrane, internal proteins, nucleus, and mitochondria) displace the water media within a ~100 nm thin region above the surface of the PC. PCEM uses a modification to the illumination and imaging paths of a conventional bright field microscope to divide a microscope field of view into 0.5 ïm pixels, in which the PC resonant spectrum is recorded, and the peak wavelength value (PWV) is determined to generate a biosensor image.
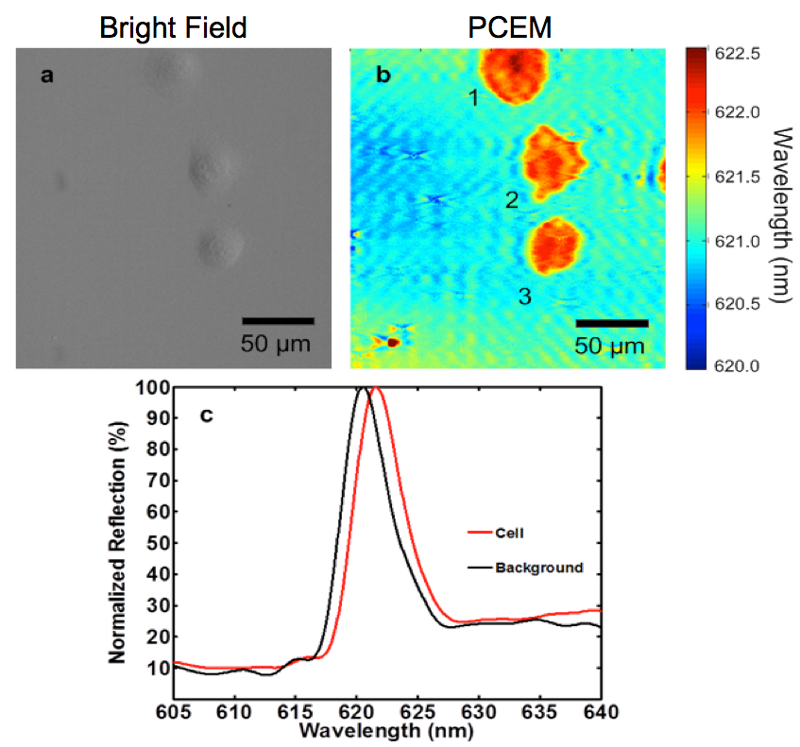
Figure 2:(a) Bright field and (b) PWV imaging of Panc-1 cells attached to the PC surface. Cells were seeded onto a fibronectin-coated sensor and allowed to incubate for 2 hours before imaging. Lamellipodial extensions are visible, especially from cell 2, demonstrating the ability of PCEM to resolve regional differences in single-cell attachment. Darker shading indicates regions of higher protein concentration, and is present in regions near the boundary of lamellipodia formation, consistent with the creation of actin bundles. (c) Representative regions of cellular attachment. Selected areas of the PWV image from beneath a cell show the PWV shift of a typical Panc-1 cell is ~1.0 nm, and consistent throughout the entire spectrum at those locations. Reproduced with permission from [1].
As shown in Figure 2, not only does PCEM easily measure the attachment of individual cells, it also measures the spatial distribution of cell attachment strength within a cell. Our PCEM microscope incorporates an integrated cell incubator, and because no cytotoxic stains or photobleachable fluorescent dyes are used, we can monitor cells over substantial periods of time. In fact, we have used PCEM to generate the first “movies” of stem cell attachment to a surface, and the evolution of the attachment footprint during chemotaxis up a chemical gradient. Biologists are developing a more sophisticated understanding of how cell membrane interactions with surfaces, chemical stimuli, and other cells are modulated, and the role of integrins, ion channels, G-coupled transmembrane proteins, and filapodia in fundamental processes such as migration, wound healing, differentiation, and apoptosis. Yet, there are few tools currently available that allow visualization and quantification of these processes. Our goal is to develop and demonstrate PCEM as a new tool for biologists to more fully understand fundamental cell processes, and a technique that can aid in measuring the interaction of cells with drugs, or other challenges to their environment.
For Further Reading
1. W. Chen, et al., “Photonic crystal enhanced microcsopy for imaging of live cell adhesion”, Analyst, Vol. 138, p. 5886, 2013
Contributor
 Brian T. Cunningham is a Professor in the Department of Electrical and Computer Engineering and the Department of Bioengineering at the University of Illinois at Urbana-Champaign, where he also serves as the Interim Director of the Micro and Nanotechnology Laboratory, and as Director of the NSF Center for Agricultural, Biomedical, and Pharmaceutical Nanotechnology. His research is in the development of biosensors and detection instruments for pharmaceutical high throughput screening, disease diagnostics, point-of-care testing, life science research, and environmental monitoring. Read more
Brian T. Cunningham is a Professor in the Department of Electrical and Computer Engineering and the Department of Bioengineering at the University of Illinois at Urbana-Champaign, where he also serves as the Interim Director of the Micro and Nanotechnology Laboratory, and as Director of the NSF Center for Agricultural, Biomedical, and Pharmaceutical Nanotechnology. His research is in the development of biosensors and detection instruments for pharmaceutical high throughput screening, disease diagnostics, point-of-care testing, life science research, and environmental monitoring. Read more







 Mary Capelli-Schellpfeffer, MD, MPA, is Medical Director of Loyola University Health System's Occupational Health Services, and Associate Professor, Department of Medicine, Loyola University Chicago Stritch School of Medicine. Dr. Mary Capelli-Schellpfeffer guides Loyola's occupational medicine programs.
Mary Capelli-Schellpfeffer, MD, MPA, is Medical Director of Loyola University Health System's Occupational Health Services, and Associate Professor, Department of Medicine, Loyola University Chicago Stritch School of Medicine. Dr. Mary Capelli-Schellpfeffer guides Loyola's occupational medicine programs.  Brian T. Cunningham is a Professor in the Department of Electrical and Computer Engineering and the Department of Bioengineering at the University of Illinois at Urbana-Champaign, where he also serves as the Interim Director of the Micro and Nanotechnology Laboratory, and as Director of the NSF Center for Agricultural, Biomedical, and Pharmaceutical Nanotechnology. His research is in the development of biosensors and detection instruments for pharmaceutical high throughput screening, disease diagnostics, point-of-care testing, life science research, and environmental monitoring.
Brian T. Cunningham is a Professor in the Department of Electrical and Computer Engineering and the Department of Bioengineering at the University of Illinois at Urbana-Champaign, where he also serves as the Interim Director of the Micro and Nanotechnology Laboratory, and as Director of the NSF Center for Agricultural, Biomedical, and Pharmaceutical Nanotechnology. His research is in the development of biosensors and detection instruments for pharmaceutical high throughput screening, disease diagnostics, point-of-care testing, life science research, and environmental monitoring.  Aniruddha Datta received the B. Tech degree in Electrical Engineering from IIT Kharagpur in 1985, the M.S.E.E. degree from Southern Illinois University, Carbondale in 1987 and the M.S. (Applied Mathematics) and Ph.D. degrees from the University of Southern California in 1991. In August 1991, he joined the Department of Electrical and Computer Engineering at Texas A&M University where he is currently the J. W. Runyon, Jr. '35 Professor II.
Aniruddha Datta received the B. Tech degree in Electrical Engineering from IIT Kharagpur in 1985, the M.S.E.E. degree from Southern Illinois University, Carbondale in 1987 and the M.S. (Applied Mathematics) and Ph.D. degrees from the University of Southern California in 1991. In August 1991, he joined the Department of Electrical and Computer Engineering at Texas A&M University where he is currently the J. W. Runyon, Jr. '35 Professor II.