Reverse Engineering the Brain
By Simon Schultz
Simon Schultz, Director of the Centre for Neurotechnology and Reader in Neurotechnology in the Department of Bioengineering, Imperial College London, is interested in “reverse engineering” the information processing architecture of the brain. His research investigations focus on the basic principles of information processing in cortical circuits through an engineering approach, which involves both doing experiments with mice as well as the theoretical work needed to understand the data from these experiments.
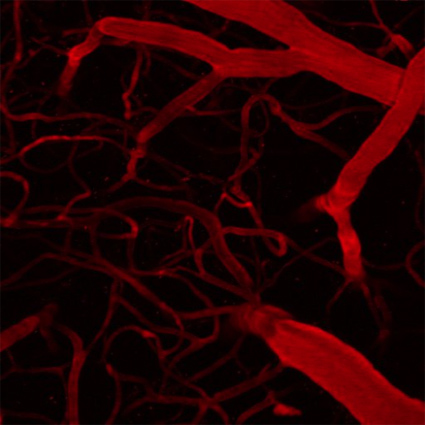
Figure 1: Two photon imaging also allows us to see the brain’s microvasculature. This helps to advance our understanding of stroke and dementia, as the healthy operation of neural circuits is critically contingent upon this network of tiny blood vessels in the brain.
Much of my work is focused on improving the technology used for studying the neural code for sensory and cognitive processes. On the experimental side, our lab uses two-photon microscopy (Figure 1), optogenetic manipulation, and electrophysiology to measure (and interfere with) patterns of neuronal activity in vivo (Figure 2). On the theoretical side, we develop new algorithms for analyzing the resulting data, and then develop models to interpret this data. Research into the functional principles of cortical circuits is crucial to understanding and also treating the effects of brain disorders. In particular, my interests center on the changes in cortical circuit properties during dementias such as Alzheimer’s disease.
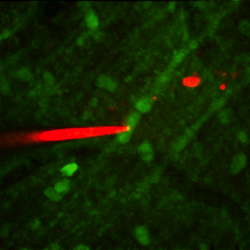
Figure 2: This image shows an electrophysiological recording (pipette filled with red dye) being made under the guidance of a two-photon microscope from the dendrites of a cerebellar Purkinje cell stained with green fluorescent dye.
Our aim is to understand how sensory information is processed by neural circuits in the mammalian cerebral and cerebellar cortices, including how it is used in elementary cognitive operations (Figure 3). Understanding how the cortical circuit processes information will help us to understand how it dysfunctions in neurodevelopmental and neurodegenerative disorders, and will also aid in the design of innovative computational devices. One of our group’s research goals is to look at repairing brain circuitry and then to develop neural technologies that interact with the nervous system. Ultimately, a range of neuroprosthetics might result from this research, which could unlock the ability to heal, or indeed replace, damaged areas of the brain.
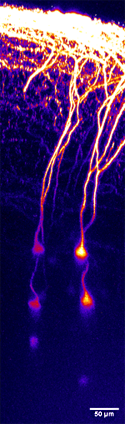
Figure 3: This image shows pyramidal neurons in layer 5 of the mouse cortex, imaged in a living brain.
Recently, we have developed some technologies for interfacing with the nervous system that can only be used in mice because interfacing with the nervous system of a paralyzed patient, for example, is much more complex. Much of our current technology for targeting individual elements of the cortical circuit depends on genetic modification of individual neurons—in humans, this comes under the heading of “gene therapy” and needs much more research before it becomes routinely possible. Nevertheless, there are some things we can do with electrode-based technologies that overlap with areas of clinical needs such as paralysis, locked-in syndrome, or loss of a limb. All of these have relatively small patient populations, in comparison to disorders such as dementia and cardiovascular disease. Alzheimer’s disease would be a much larger population, but relatively little can be done at the moment beyond the basic research. Eventually, we hope to be able to develop memory enhancement technology, but that is beyond our capability now in terms of clinical application.
To tackle dementia we need to know more basic science. This difficult problem requires using many different approaches, including neuroscience and engineering. One area that holds promise is optogenetics—a neuromodulation technique that uses light to affect neurons that have been genetically sensitized to light, while at the same time enabling the control and monitoring of activity in individual neurons in living tissue as well as the real-time study of the effects of those manipulations. Optogenetics can now be used to disturb brain activity by affecting neurons of a single type as a group. It gives us the ability to watch patterns of brain activity during certain behaviors. Also, new wavefront technology has the potential to address specific areas of neurons. All of this provides a new window on brain activity. Most of what we currently know about brain activity comes from single electrode reading techniques, but with these innovative approaches we can record thousands at once.
Advances have also been made in techniques such as patch clamping, which allows the study of single or multiple ion channels in cells, particularly excitable cells such as neurons, cardiomyocytes, muscle fibers, and pancreatic beta cells. An automated version of the technique is enabling easier and faster study of the inner workings of brain cells. For example, patch clamping has now been automated at the Massachusetts Institute of Technology (MIT), where a robot can guide the pipette into the brain, monitoring the resistance of the pipette and controlling its pressure, in order to automatically attach onto the cell membrane. We have augmented this technology by incorporating a two-photon microscope on top of the patching robot, which allows us to take a scan of an area of tissue in the living brain and instruct the robot to target a specific cell (based, for instance, on its genetic class, which we can see via a fluorescent label) for patch clamping.
Other lab research areas include inferring the times of sequences of action potentials (APs) (spike trains) from neurophysiological data, which is a key problem in computational neuroscience. The detection of APs from two-photon imaging of calcium signals offers certain advantages over traditional electrophysiological approaches, as up to thousands of spatially and immunohistochemically defined neurons can be recorded simultaneously. However, due to noise, the kinetics of fluorescent calcium sensors, and the limited sampling rates in common microscopy configurations, accurate detection of APs from calcium time series has proved to be a difficult problem.
Recently, we have developed a novel approach to the problem by making use of finite rate of innovation (FRI) theory. For calcium transients well fit by a single exponential, the problem is reduced to reconstructing a stream of decaying exponentials. Signals made of a combination of exponentially decaying functions with different onset times are a subclass of FRI signals, for which much theory has recently been developed by the signal processing community. In our research, we demonstrate the use of FRI theory to retrieve the timing of APs from calcium transient time series. This final algorithm is fast, non-iterative, and parallelizable and has been tested with both real data (obtained by simultaneous electrophysiology and multiphoton imaging of calcium signals in cerebellar Purkinje cell dendrites), and surrogate data.
Selected Publications:
- J. Oñativia, S. R. Schultz and P.-L. Dragotti. “A Finite Rate of Innovation algorithm for fast and accurate spike detection from two-photon calcium imaging,” Journal of Neural Engineering, vol. 10 046017, no. 4, 2013.
- N. Grossmann, V. Simiaki, C. Martinet, C. Toumazou, S. R. Schultz, and K. Nikolic, “The spatial pattern of light determines the kinetics and modulates backpropagation of optogenetic action potentials.” Journal of Computational Neuroscience, vol. 34, no. 3, pp. 477-88, 2013. doi: 10.1007/s10827-012-0431-7.
- E. Phoka, M. Wildie, S. R. Schultz, and M. Barahona, “Sensory experience modifies spontaneous state dynamics in a barrel cortical circuit model,” Journal of Computational Neuroscience, vol. 33, pp. 323-339, 2012.
- A. B. Saleem, K.D. Longden, D. Schwyn, H. G. Krapp, and S. R. Schultz, “Bimodal optomotor response to plaids in blowflies: Mechanisms of component-selectivity and evidence for pattern-selectivity,” Journal of Neuroscience, vol. 32, no. 5, pp. 1634-1642, 2012.
- M. T. Schaub and S. R. Schultz.,”The Ising Decoder: Reading out the activity of large neural ensembles,” Journal of Computational Neuroscience, vol. 32, no.1, pp. 101-118, 2012.
Contributor
 Simon Schultz is Director of the Centre for Neurotechnology at Imperial College London. After receiving degrees in Electrical & Computer Systems Engineering and Physics, he earned his PhD in the area of computational neuroscience under Prof. Edmund Rolls at Oxford. He conducted postdoctoral research in experimental neuroscience at New York University and University College London.
Simon Schultz is Director of the Centre for Neurotechnology at Imperial College London. After receiving degrees in Electrical & Computer Systems Engineering and Physics, he earned his PhD in the area of computational neuroscience under Prof. Edmund Rolls at Oxford. He conducted postdoctoral research in experimental neuroscience at New York University and University College London.
Read more







 Steve Furber CBE FRS FREng is Imperial College London (ICL) Professor of Computer Engineering in the School of Computer Science at the University of Manchester, UK. There, he leads research into asynchronous and low-power systems and, more recently, neural systems engineering.
Steve Furber CBE FRS FREng is Imperial College London (ICL) Professor of Computer Engineering in the School of Computer Science at the University of Manchester, UK. There, he leads research into asynchronous and low-power systems and, more recently, neural systems engineering. Simon Schultz is Director of the Centre for Neurotechnology at Imperial College London. After receiving degrees in Electrical & Computer Systems Engineering and Physics, he earned his PhD in the area of computational neuroscience under Prof. Edmund Rolls at Oxford.
Simon Schultz is Director of the Centre for Neurotechnology at Imperial College London. After receiving degrees in Electrical & Computer Systems Engineering and Physics, he earned his PhD in the area of computational neuroscience under Prof. Edmund Rolls at Oxford.