Smart Implants for Surgery
By Henry Ip and Guang-Zhong Yang
Increasingly, “smart” implants which incorporate information processing components are coming into use. Long-term monitoring is now possible, as new power sources emerge.
Smart implants with integrated information processing play an important role in modern medicine in the form of neural prosthetics (see [1] for a recent review). The most widely implanted smart devices being the pacemaker/defibrillator [2] and the cochlea implant for the profoundly deaf [3]. Recently there is a shift in landscape in the use of smart medical implants for long term and transient post-operative monitoring. The implanted device provides early detection of adverse events thus minimising post-operative complication by ensuring prompt instigation of appropriate treatments. This is in contrast to the “wait and see” approach of traditional post-operative care pathways (Fig 1).
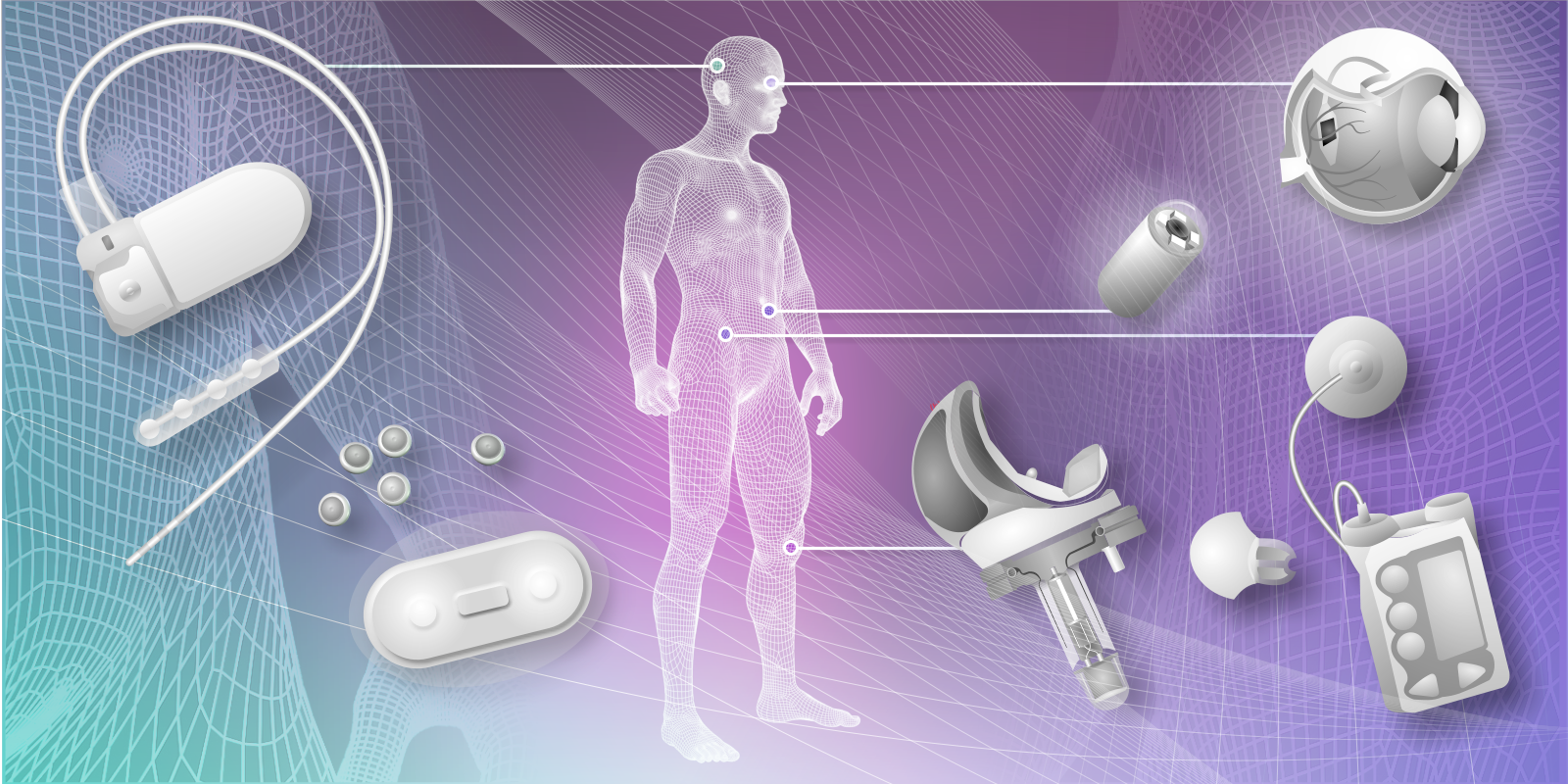
Figure 1: The range of implantable/ingestible sensors (ranging from neuro-stimulator (NeuroPace), retinal implant, smart pill (Proteus), to smart knee prosthesis) already in use or being developed for patient monitoring (for further details, see http://www.springer.com/computer/hci/book/978-1-4471-6373-2, image courtesy of RD Merrifield).
The majority of implantable devices with sensing and information encoding/processing capabilities are “active” devices – meaning a power source is required for device operation. This is contrary to some passive implantable sensors (e.g. [4]) which externally measureable electrical properties of the device are modulated by the sensing target. The active implants, however, typically offers more accurate sensing capabilities and engineered to work with a range of sensors as well as provides a means of stimulation when required. The provision of a power source, while enabling the many possible features of an active implant, bears a great engineering challenge. State-of-art power harvesting devices operating within the body to harvest energy from heat and blood flow provides limited power up to a maximum of 1-200 micro Watts. One of the proven ways of overcoming the technical challenge of conveying larger power into an implanted device is via magnetic field power transmission [5]. This method is adopted by a number of commercial cochlea implants mostly due to the low attenuation of magnetic fields inside tissues. To this end, an external device is used to generate a magnetic field. The implanted device consists of magnetic field sensing “coils” and associated circuitry to convert parts of the energy from the externally applied magnetic field to power the active implant. The coils used in the power transceivers are effectively air-core inductors and the process of wireless power transmission operates under inductive coupling principles. This is widely used commercially in the areas of RFID smart cards. Inductive coupling also facilitates bi-directional data transmission, which is often the more power consuming part of a smart implant with the other significant power drain comes from any stimulation circuits. The power requirement of data transmission and stimulation is often not met by ambient in-body energy harvesting methods making inductive wireless/non-contact powering an attractive feature.
One of the key design constraints of an implant is the device size. Integrated circuit and other miniaturisation technology has enabled a large number of sensing and stimulation circuitry to be implemented in a very small area, typically in the mm scale with some devices measuring only a few hundred microns on each side. However, the size of an inductively powered implant is often determined by the size of the inductor coil as the available power is limited by reduced coil size. The distance of efficient power transmission also reduces with coil size. For comparison, a typical smart card device have utilise coils in the order of centimeters. Coils in these devices are typically made of printed materials enabling low cost production. On the other hand, implants are expected to be as miniaturised as possible to improve patient comfort. The use of Ferrite beads in the implant coil greatly improves its the magnetic field power harvesting capability by concentrating the received field within the Ferrite, which in turn is sensed by the coil around the ferrite core. Recent experiments in implant prototypes with Ferrite core coils show promising results of receiving power in the mW range over a few centimeters over a modest power source (Fig. 2).
Fig.2. An implant coil for receiving wireless power.
Typical building blocks of an active implant consist of integrated circuitry for sensing, stimulation, and data transmission. Sensing circuitry, while normally operates on a smaller power budget (~uW) compared to circuits for stimulation and data communications, requires special attention on the design of the power supply. For inductively powered implants, the power recovery circuit converts the high frequency magnetic field into a steady voltage suitable for use by the rest of the implant. Since sensing circuits typically handles very small signals from the sensors, it is more prone to signal corruption when the power supply voltage is not sufficiently filtered. So despite promising power budget from ferrite core implant coils, a systems approach must be adopted to meet sensing, power, and data transmission design targets.
For Further Reading
1. J Ohta, T Tokuda, K Sasagawa, T Noda, “Implantable CMOS Biomedical Devices,” Sensors, vol. 9, pp 9073-9093, 2009.
2. M Glikson, P A Friedman, “The implantable cardioverter defibrillator,” The Lancet, Vol. 357, Issue 9262, pp 1107-1117, April 2001.
3. F-G Zeng, S Rebcher, W Harrison, X Sun, H Feng, “Cochlear implants: system design, integration, and evaluation,” IEEE Rev. Biomedical Engineering, Vol. 1, pp 115-142, 2008.
4. H Murphy, M R Bahmanyar, A Borghi, C N McLeod, M Navaratnarajah, M H Yacoub, C Toumazou, “Continuous in vivo blood pressure measurements using a fully implantable wireless SAW sensor,” Biomed Microdevices, 15:737-749, 2013.
5. M V Paemel, “High-efficiency transmission for medical implants,” IEEE Solid-State Circuits Magazine, pp47-59, Winter 2011.
Contributors
 Henry Ip is a post-doctoral research fellow at the Hamlyn Centre, Imperial College London, UK. He is specialised in bio-inspired architectures for low-power Application Specific Integrated Circuits (ASICs), miniature amperometric ASIC arrays for subcellular electrochemical recordings and nano-power continuous-time filters. Read more
Henry Ip is a post-doctoral research fellow at the Hamlyn Centre, Imperial College London, UK. He is specialised in bio-inspired architectures for low-power Application Specific Integrated Circuits (ASICs), miniature amperometric ASIC arrays for subcellular electrochemical recordings and nano-power continuous-time filters. Read more
 Guang-Zhong Yang is director and co-founder of the Hamlyn Centre, Imperial College London, UK. Professor Yang’s main research interests are in medical imaging, sensing and robotics. He is a Fellow of the Royal Academy of Engineering, fellow of IEEE, IET, AIMBE and a recipient of the Royal Society Research Merit Award and listed in The Times Eureka ‘Top 100’ in British Science. Read more
Guang-Zhong Yang is director and co-founder of the Hamlyn Centre, Imperial College London, UK. Professor Yang’s main research interests are in medical imaging, sensing and robotics. He is a Fellow of the Royal Academy of Engineering, fellow of IEEE, IET, AIMBE and a recipient of the Royal Society Research Merit Award and listed in The Times Eureka ‘Top 100’ in British Science. Read more







 Surapa Thiemjarus is a researcher from the National Electronics and Computer Technology Center (NECTEC), National Science and Technology Development Agency (NSTDA), Thailand. She received her PhD degree in Computing from Imperial College, UK. Her research interests include context-aware and pervasive sensing, body sensor networks and applications, sensor fusion, machine learning, pattern recognition, sound and signal processing, and assistive technologies.
Surapa Thiemjarus is a researcher from the National Electronics and Computer Technology Center (NECTEC), National Science and Technology Development Agency (NSTDA), Thailand. She received her PhD degree in Computing from Imperial College, UK. Her research interests include context-aware and pervasive sensing, body sensor networks and applications, sensor fusion, machine learning, pattern recognition, sound and signal processing, and assistive technologies.  Jing Liu is currently working as a short-term research assistant in Joint Research Center for Biomedical Engineering at the Chinese University Hong Kong (CUHK), Shatin, N.T., Hong Kong. Meanwhile, she is a senior student of Wuhan University majoring in Computer Science and she is going to pursue her Ph.D. degree in Biomedical Engineering at CUHK in the autumn of 2014. Her research interests include wearable medical device and physiological modeling.
Jing Liu is currently working as a short-term research assistant in Joint Research Center for Biomedical Engineering at the Chinese University Hong Kong (CUHK), Shatin, N.T., Hong Kong. Meanwhile, she is a senior student of Wuhan University majoring in Computer Science and she is going to pursue her Ph.D. degree in Biomedical Engineering at CUHK in the autumn of 2014. Her research interests include wearable medical device and physiological modeling.  Xiao-Rong Ding is currently pursuing her Ph.D. degree in Biomedical Engineering at the Joint Research Centre for Biomedical Engineering of The Chinese University of Hong Kong, Shatin, N.T., Hong Kong. Her research interests include wearable medical devices, CVD health informatics and physiological modelling.
Xiao-Rong Ding is currently pursuing her Ph.D. degree in Biomedical Engineering at the Joint Research Centre for Biomedical Engineering of The Chinese University of Hong Kong, Shatin, N.T., Hong Kong. Her research interests include wearable medical devices, CVD health informatics and physiological modelling.  Ni Zhao is currently an assistant professor at the Department of Electronic Engineering at the Chinese University of Hong Kong, Shatin, N.T., Hong Kong. She received her Ph.D. degree in Physics from the University of Cambridge (UK) in 2008. Her research interest covers optoelectronic, electronic and electrochemical devices based on organic and nanostructured materials; spectroscopic characterization of the physical processes in nanostructured thin films, structures, and devices.
Ni Zhao is currently an assistant professor at the Department of Electronic Engineering at the Chinese University of Hong Kong, Shatin, N.T., Hong Kong. She received her Ph.D. degree in Physics from the University of Cambridge (UK) in 2008. Her research interest covers optoelectronic, electronic and electrochemical devices based on organic and nanostructured materials; spectroscopic characterization of the physical processes in nanostructured thin films, structures, and devices.  Yuan-Ting Zhang is currently the Director of Joint Research Center for Biomedical Engineering and Professor of Department of Electronic Engineering at the Chinese University of Hong Kong (CUHK), Hong Kong, China. He serves concurrently the Director of the Key Lab for Health Informatics of the Chinese Academy of Sciences (HICAS) at SIAT, Shenzhen, China. He is the founder and the first Director of the SIAT Institute of Biomedical and Health Engineering under the Chinese Academy of Sciences. His research spans several fields including wearable medical devices, flexible biosensors, BSN security, neuro-physiological modeling, CVD health informatics, and m-u-p-Heath technologies.
Yuan-Ting Zhang is currently the Director of Joint Research Center for Biomedical Engineering and Professor of Department of Electronic Engineering at the Chinese University of Hong Kong (CUHK), Hong Kong, China. He serves concurrently the Director of the Key Lab for Health Informatics of the Chinese Academy of Sciences (HICAS) at SIAT, Shenzhen, China. He is the founder and the first Director of the SIAT Institute of Biomedical and Health Engineering under the Chinese Academy of Sciences. His research spans several fields including wearable medical devices, flexible biosensors, BSN security, neuro-physiological modeling, CVD health informatics, and m-u-p-Heath technologies.  Henry Ip is a post-doctoral research fellow at the Hamlyn Centre, Imperial College London, UK. He is specialised in bio-inspired architectures for low-power Application Specific Integrated Circuits (ASICs), miniature amperometric ASIC arrays for subcellular electrochemical recordings and nano-power continuous-time filters.
Henry Ip is a post-doctoral research fellow at the Hamlyn Centre, Imperial College London, UK. He is specialised in bio-inspired architectures for low-power Application Specific Integrated Circuits (ASICs), miniature amperometric ASIC arrays for subcellular electrochemical recordings and nano-power continuous-time filters.  Guang-Zhong Yang is director and co-founder of the Hamlyn Centre, Imperial College London, UK. Professor Yang's main research interests are in medical imaging, sensing and robotics. He is a Fellow of the Royal Academy of Engineering, fellow of IEEE, IET, AIMBE and a recipient of the Royal Society Research Merit Award and listed in The Times Eureka 'Top 100' in British Science.
Guang-Zhong Yang is director and co-founder of the Hamlyn Centre, Imperial College London, UK. Professor Yang's main research interests are in medical imaging, sensing and robotics. He is a Fellow of the Royal Academy of Engineering, fellow of IEEE, IET, AIMBE and a recipient of the Royal Society Research Merit Award and listed in The Times Eureka 'Top 100' in British Science.