A system for active compensation of motion artifacts in non-contact ECG sensing
By Guochen Peng and Mark F. Bocko
Non-contact ECG monitoring is an attractive option for a number of applications such as long-term ambulatory health monitoring. Subject-electrode relative motion is a source of spurious signals and significant signal distortion. Our group has developed a non-contact ECG sensing system including electrodes, interface electronics and a signal-processing unit that work together to address this issue. The subject-to-electrode distance is continuously monitored by a secondary sensing circuit that uses the ECG electrodes, which serve as both the primary ECG readout and the secondary subject-to-electrode distance readout. Experimental results demonstrate the effectiveness of the compensation scheme.
Traditional electrocardiogram (ECG) adhesive sensor electrodes provide a stable, low impedance signal source that enables low noise measurement of ECG signals, but the wires and adhesives are a considerable inconvenience and discomfort to the user, especially in long term personal health monitoring scenarios. The development of dry-contact and non-contact electrodes in conjunction with wireless technology addresses these issues.
Dry-contact electrodes are formfitting and can take a form of body-worn sticker [1], chest strap [2], fingertip plate [3], and so on. However, these devices still require good contact with the subject’s skin for reliable sensing and may be cumbersome for some individuals. An alternative is to use non-contact sensing which was successfully demonstrated by Prance [4] and Ueno [5]. The development of non-contact sensors that may be built into clothing, examination tables, beds, or other furniture is an attractive option. However, there are significant practical challenges and a good deal of on-going research, for example in the development of suitable preamplifiers [6, 7], elimination of common-mode electromagnetic interference coupled from power mains [8], triboelectrically generated charge from the rubbing of the electrodes on the subject’s clothing [9], and modulation of the bioelectric potential signals from motion-related source impedance changes [10,11].
Changes in the source capacitance due to the relative motion of the electrodes to the subject lead to the modulation of both the signal of interest as well as the above-mentioned sources of interference and noise, which may have significant power within the signal band of interest. This effect may be large enough in practical scenarios to completely obscure the ECG signal. One solution [11] is to directly monitor the subject-electrode motion with a proximity capacitive sensor using the ECG electrodes and then to electronically compensate for the changes in the signal source capacitance. By employing an additional static charge sensor, the ECG signal can be retrieved from the corrupted measurements.
In Figure 1(a) an ECG electrode and guard ring are shown. The capacitance from the ECG electrode to the subject is a function of the subject-electrode gap and the electrode area. At the same time, the capacitance between the guard electrode and the ECG electrode is also a function of the sensor-to-subject gap as well as other geometric factors [12]. By monitoring the electrode to guard ring capacitance, the ECG electrode-to-subject capacitance Cs can be directly inferred.
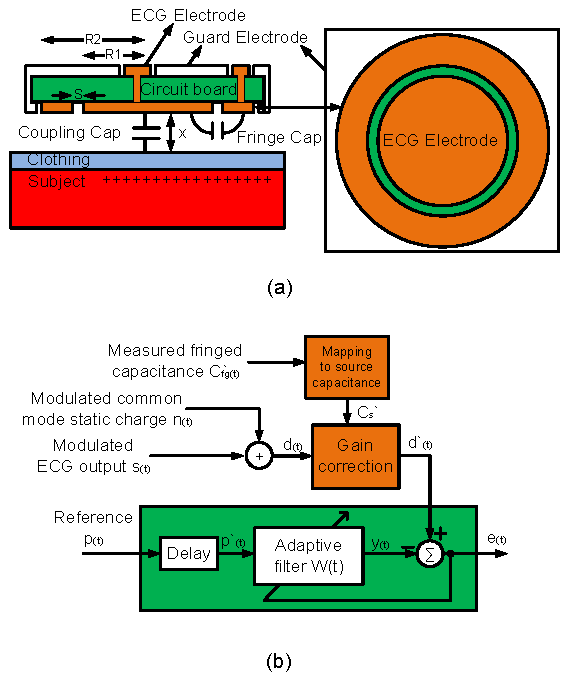
Figure 1: (a) The ECG Electrode is surrounded by a guard electrode to reduce stray capacitive coupling to external electric fields. The fringe capacitance between the guard electrode and the ECG electrode is a function of the sensor-to-subject distance and thus may be employed to infer the ECG electrode-to-subject capacitance. (b) Conceptual diagram of ECG correction method including the adaptive filtering system.
The corrupted ECG signal has two additive components [10,11]: the common-mode static charge signal and the ECG signal; both are also modulated by changes in the source capacitance. Therefore, the artifacts can be divided into additive and modulation components. Using the auxiliary displacement measurement enables gain correction of the ECG signal employing an adaptive filtering algorithm. Referring to Figure 1(b), the adaptive filtering module includes two subsystems for processing three primary inputs; the measured fringe capacitance C’fg, the distorted ECG signal d, and the static charge reference signal p. The system first makes the gain correction and then eliminates the additive static common-mode interference.
The first step in the adaptive filtering process is to measure the fringe capacitance C’fg and convert this quantity into an equivalent source capacitance value. The source capacitance C‘ can be used to actively gain-correct the distorted ECG signal. After gain correction is complete, the residual signal d‘ contains two additive terms: a purely linear scaled ECG signal s’ and static charge interference n‘. Therefore, the measured reference p should be highly correlated with the common-mode static charge n‘.
The full architecture for the non-contact ECG sensor, the proximity sensor, and static charge electrometer is shown in Figure 2(a). The subject is capacitively sensed through clothing with a sensor electrode. The input terminal of the proximity sensor bridge circuit is coupled to secondary electrode, while the ECG charge preamplifier is coupled through a low pass filter to the primary ECG electrode. The use of a low-pass filter removes the high frequency signals (100kHz) employed by the proximity sensor. A high impedance electrometer serves as a static charge monitor for the input reference p.

Figure 2: (a) The proximity sensor (upper signal chain), the ECG readout (middle signal chain), and the static charge electrometer are digitized and the inferred changes in ECG electrode capacitance (from the proximity sensor data) are employed to apply gain compensation to the ECG signal. (b) ECG compensation illustration. The lower panel shows the ECG electrode capacitance and distance between electrode and the subject inferred from the fringe capacitance measurement. In the middle panel the uncorrected raw ECG signal is shown, while in the upper trace the corrected ECG signal is shown. The amount of distortion in this test was minimal; however in the corrected ECG signal the R peak height is more consistent.
The circuit of Figure 2(a) was implemented with a mock-up of the signal source by applying a time-varying voltage resembling an ECG waveform to an aluminum enclosure covered in textile. Random motion of the sensor was induced with a translation stage and track. The ECG and proximity sensor outputs were separately collected for signal reconstruction. The results in Figure 2(b) illustrate the effectiveness of the compensation method. One of the key features of this scheme is that the guard-ring proximity sensing circuit and the ECG sensor operate simultaneously without mutual interference because the AC bridge circuit that is used in the proximity sensor readout operates at 100 kHz, which is far above the bandwidth of interest in the ECG signal channel and thus is greatly attenuated by a low-order filter.
In summary, the signal reconstruction method and system described is capable of monitoring the electrode-to-subject distance while providing the ECG output using the same electrode. In ongoing work our group is developing a system-on-chip ASIC that implements the systems described here to enable a higher degree of integration and a more compact form factor.
Acknowledgements
We wish to thank Dr. Mark Sterling, Dr. John Liobe, and Dr. Zeljko Ignjatovic, for their assistance. This research was supported by Blue Highway LLC, Welch-Allyn Inc., the New York State Foundation for Science, Technology, and Innovation, and the Eunice Kennedy Shriver National Institute of Child Health Development Grant R01HD060789.
For Further Reading
1. D.H. Kim, et al., “Epidermal electronics”. Science, 2011. 333(6044): p. 838-43.
2. http://www.underarmour.com/shop/us/en/armour39
3. http://www.alivecor.com/home
4. A.J. Clippingdale, et al., “Ultrahigh Impedance Capacitively Coupled Heart Imaging Array.” Review of Scientific Instruments, 1994. 65(1): p. 269-270.
5. A. Ueno, et al., “Capacitive sensing of electrocardiographic potential through cloth from the dorsal surface of the body in a supine position: A preliminary study.” IEEE Transactions on Biomedical Engineering, 2007. 54(4): p. 759-766.
6. G. Peng, Z. Ignjatovic, and M. Bocko, “Preamplifiers for non-contact capacitive biopotential measurements,” in EMBC, 2013, IEEE, 3-7 Jul. 2013.
7. G. Peng and M. Bocko, “A low noise, non-contact capacitive cardiac sensor,” in EMBC, 2012, IEEE, 28 Aug. -1 Sept. 2012.
8. G. Peng and M. Bocko, “Non-contact ecg sensing employing gradiometer electrodes,” IEEE Transactions on Biomedical Engineering, vol. 60, no. 1, pp. 179-183, 2013.
9. G. Peng, M. Sterling, and M. Bocko, “Non-contact biosensor electrodes for ESD reduction,” in Sensors, 2013, IEEE, 3-6 Nov 2013.
10. J. Ottenbacher and S. Heuer, “Motion artefacts in capacitively coupled ecg electrodes,” in World Congress on Medical Physics and Biomedical Engineering, September 7 – 12, 2009, Munich, Germany, ser. IFMBE Proceedings, R. Magjarevic, O. Dossel, and W. C. Schlegel, Eds. Springer Berlin Heidelberg, 2010, vol. 25/4, pp. 1059-1062.
11. G. Peng and M. Bocko, “Non-contact, Capacitive ECG Employing Signal Compensation,” in IEEE Biomedical Circuits and Systems Conference (BioCAS), 2013, IEEE, 31 Oct. -2 Nov. 2013.
12. Z. Chen and R. Luo, “Design and implementation of capacitive proxim- ity sensor using microelectromechanical systems technology,” IEEE Transactions on Industrial Electronics, vol. 45, no. 6, pp. 886-, Dec 1998.
Contributors
 Guochen (Benjamin) Peng is currently working toward the Ph.D. degree in Electrical and Computer Engineering at the University of Rochester. His research interests include VLSI mixed signal design, biomedical sensor electronic devices and signal processing, and precision measurements. Read more
Guochen (Benjamin) Peng is currently working toward the Ph.D. degree in Electrical and Computer Engineering at the University of Rochester. His research interests include VLSI mixed signal design, biomedical sensor electronic devices and signal processing, and precision measurements. Read more
 Mark F Bocko is Mark F Bocko received the Ph.D. degree in physics from the University of Rochester, Rochester, NY. Currently, he is Full Professor of the Department of the Electrical and Computer Engineering Department, University of Rochester. His research interests span a number of areas, including sensors and integrated sensor systems, audio and music signal processing, precision measurements, superconducting electronics quantum noise, and quantum computing. Read more
Mark F Bocko is Mark F Bocko received the Ph.D. degree in physics from the University of Rochester, Rochester, NY. Currently, he is Full Professor of the Department of the Electrical and Computer Engineering Department, University of Rochester. His research interests span a number of areas, including sensors and integrated sensor systems, audio and music signal processing, precision measurements, superconducting electronics quantum noise, and quantum computing. Read more







 Mohamad Sawan received the Ph.D. degree in Electrical Engineering from Sherbrooke University, Canada. He joined Polytechnique Montréal in 1991, where he is currently a Professor of microelectronics and biomedical engineering. His interests are the mixed-signal circuits and Microsystems.
Mohamad Sawan received the Ph.D. degree in Electrical Engineering from Sherbrooke University, Canada. He joined Polytechnique Montréal in 1991, where he is currently a Professor of microelectronics and biomedical engineering. His interests are the mixed-signal circuits and Microsystems.  Camilla Baj-Rossi is currently pursuing the Ph.D. degree at the Laboratory of Integrated Systems, EPFL. Her current research interests include the development of a point-of-care biosensor based on multi-walled carbon-nanotubes and cytochrome P450 to detect drugs in biological fluids.
Camilla Baj-Rossi is currently pursuing the Ph.D. degree at the Laboratory of Integrated Systems, EPFL. Her current research interests include the development of a point-of-care biosensor based on multi-walled carbon-nanotubes and cytochrome P450 to detect drugs in biological fluids.  Giovanni De Micheli received the Ph.D. degree in electrical engineering and computer science from the University of California, Berkeley, CA, USA. He is a Professor and the Director of the Institute of Electrical Engineering and the Integrated Systems Center, École Polytechnique Fédérale de Lausanne, Lausanne, Switzerland. He is the Program Leader of the Nano-Tera.ch Program. His current research interests include emerging technologies, networks on chips and 3-D integration, and heterogeneous platform design, including electrical components and biosensors, as well as data processing of biomedical information.
Giovanni De Micheli received the Ph.D. degree in electrical engineering and computer science from the University of California, Berkeley, CA, USA. He is a Professor and the Director of the Institute of Electrical Engineering and the Integrated Systems Center, École Polytechnique Fédérale de Lausanne, Lausanne, Switzerland. He is the Program Leader of the Nano-Tera.ch Program. His current research interests include emerging technologies, networks on chips and 3-D integration, and heterogeneous platform design, including electrical components and biosensors, as well as data processing of biomedical information.  Sandro Carrara is a Scientist and Lecturer with École Polytechnique Fédérale de Lausanne, Lausanne, Switzerland. He is a former Professor of optical and electrical biosensors at the Department of Electrical Engineering and Biophysics, University of Genoa, Genoa, Italy, and Former Professor of nanobiotechnology at the University of Bologna, Bologna, Italy. His current research interests include electrical phenomena of nano bio structured films and complementary metal-oxide-semiconductor design of biochips based on proteins and DNA.
Sandro Carrara is a Scientist and Lecturer with École Polytechnique Fédérale de Lausanne, Lausanne, Switzerland. He is a former Professor of optical and electrical biosensors at the Department of Electrical Engineering and Biophysics, University of Genoa, Genoa, Italy, and Former Professor of nanobiotechnology at the University of Bologna, Bologna, Italy. His current research interests include electrical phenomena of nano bio structured films and complementary metal-oxide-semiconductor design of biochips based on proteins and DNA.  Guochen (Benjamin) Peng is currently working toward the Ph.D. degree in Electrical and Computer Engineering at the University of Rochester. His research interests include VLSI mixed signal design, biomedical sensor electronic devices and signal processing, and precision measurements.
Guochen (Benjamin) Peng is currently working toward the Ph.D. degree in Electrical and Computer Engineering at the University of Rochester. His research interests include VLSI mixed signal design, biomedical sensor electronic devices and signal processing, and precision measurements.  Mark F Bocko received the Ph.D. degree in physics from the University of Rochester, Rochester, NY. Currently, he is Full Professor of the Department of the Electrical and Computer Engineering Department, University of Rochester. His research interests span a number of areas, including sensors and integrated sensor systems, audio and music signal processing, precision measurements, superconducting electronics quantum noise, and quantum computing.
Mark F Bocko received the Ph.D. degree in physics from the University of Rochester, Rochester, NY. Currently, he is Full Professor of the Department of the Electrical and Computer Engineering Department, University of Rochester. His research interests span a number of areas, including sensors and integrated sensor systems, audio and music signal processing, precision measurements, superconducting electronics quantum noise, and quantum computing.