NeuroChip – Platform for Neuronal Injury, Repair and Regeneration
By Nitish V. Thakor
Microfabrication techniques allow the creation of a platform where neurons and related cells can be grown. Injury, repair, and regeneration processes can thereby be studied outside the body.
The nervous system is possibly the world’s most complex structure, with 100 billion plus neuronal cells, a vast network of axonal interconnections, and a thousand-fold more synaptic connections. Accompanying the neurons are the interneurons, glial cells, oligodendrocytes and the Schwann cells. Therefore understanding and constructing such networks remains a daunting task. A top-down approach is the Human Connectome Project (http://www.humanconnectomeproject.org/), using various imaging and histological techniques reconstructing the nervous system architecture from mm scale (using various MRI techniques) to nm scale (electron microscopy and histology). An alternative approach is to reconstruct the nervous system bottom-up, to “reverse engineer” the brain (this is one of the grand challenges identified by the National Academy of Engineering (http://www.engineeringchallenges.org). One way to address this challenge of reverse engineering the brain is to build it up from its cellular components, eventually constructing the neuronal network connections. The first step in this direction is taken by the development of a platform called the “NeuroChip”.
The NeuroChip is designed using the principles of microfabrication (http://en.wikipedia.org/wiki/Microfabrication). Micrometer scale structures are created with the help of polymeric materials to grow, contain and pattern the neurons and other accompanying cells. The most popular microfabrication technology is known as soft lithography [1]. “Soft” materials such as polymers like PDMS and gels, known as hydrogels, are used as substrates. Patterning using the photolithography method is combined with chemical etching of silicon or glass to create the microstructures. The essential approach, shown Figure 1, is to create a region where neurons can be grown. However, neurons are quite motile and seek to grow their axons and dendrites and, when possible, form synapses and interconnects with other cells. Without the help of any mechanical or chemical cues, their growth in vitro is quite undirected. Therefore, it becomes essential to localize the neurons and guide the axons to their targets. This is accomplished by etching, using photolithography for optical patterning of microstructures and chemical etching to etch or release the desired microstructures. Figure 1 shows a micrograph of a microchamber with compartments that separate neuronal cell bodies and channels that guide axons. The accompanying speeded up time lapse Movie Clip 1 (courtesy N. L. Jeon) shows growth of axons in the NeuroChip.
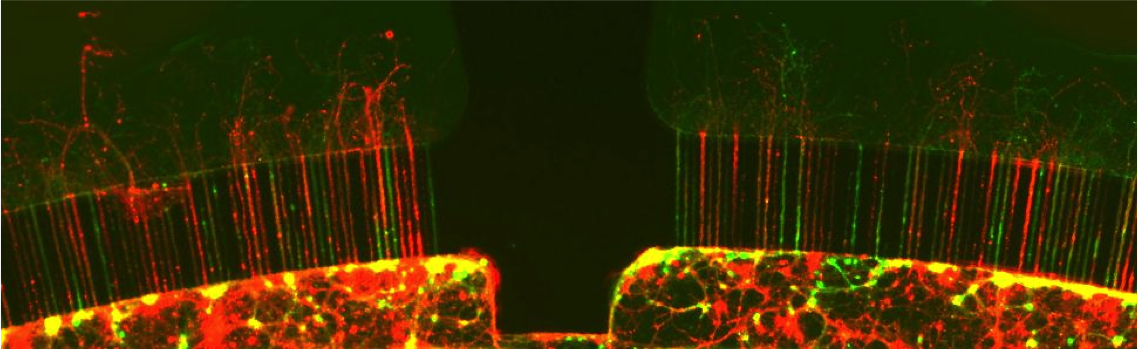
Figure 1
Movie Clip 1
Ongoing research focuses on building a multi-compartment system for separating neuronal cells bodies, or soma, from their axonal or dendritic components [2]. Taking this idea forward, axons are patterned through small micro sized grooves to their targets, which may be other neurons, to form synapses; or other cells, such myoblasts (muscle cells), to form neuromuscular junctions as seen in Movie Clip 2(courtesy I. H. Yang).
Movie Clip 2
This multicompartment design to grown neurons and interconnect them with other cells is indeed a popular area of research. The long term goal is to construct neural networks in vitro for studying the properties of cell networks as well as to demonstrate what is called a “bionic” interface. In addition, our multi-compartment system can also co-culture diverse set of cells. One important project is to couple neuronal cells to oligodendrocytes, as a model of myelination of axons in the chip [3]. Oligodendrocytes, or Schwann cells in the spinal cord, wrap on to produce what is known as myelination, essentially a punctuated series of insulation areas to promote speedier conduction in axons.
Our NeuroChip has been used in several applications; an important timely one is the study of axonal injury. Traumatic brain injury has received considerable attention in the context of sport, industrial and war related injuries. Impact injury or crush injury potentially crushes or applies shear stresses to the axon causing degeneration. The NeuroChip platform is used to study the effects of injury at single cell, single axon level, visualizing the regeneration of axon after compressive or stretch stresses as shown in Movie Clip 3 (courtesy S. Hosmane [4]).
Movie Clip 3
Injury to the spinal cord results in a number of complex changes, including damage (transection or crush) to axons, and release of factors that block their growth or affect their regeneration beyond the site of injury. This complex situation cannot be readily studied in vivo. Therefore, we created an in vitro NeuroChip platform to study the axonal injury, how its growth is impeded after injury and how subsequent regeneration can be promoted. For example, we found that inhibition of the molecule non-muscle myosin II promoted axon growth over inhibitory molecules such as CSPG [5]. Injury to the central nervous system, commonly through traumatic brain injury, leads to axonal stretch and compression. Central nervous system cells, when injured, are degraded by body’s own natural processes. For example, axons undergo what is known as Wallerian degeneration. Our NeuroChip platform has been used to observe the process of axonal degeneration and how cells known as glia degrade and digest the degenerating axons [6]. NeuroChip provides a window into the live process that go on in vivo during the injury, repair and regeneration [7]. Many spinal cord axons are also myelinated; they are covered by an insulating sheath that improves conduction. When injury occurs, this myelination is destroyed and is difficult to regenerate and wrap around repaired or regenerating axons. Ongoing in vitro research on the NeuroChip platform is attempting to develop electrical or chemical means to promote the myelination process. The NeuroChip platform, thus, allows mimicking of neuronal (and axonal) injury process and testing the strategies for repair and regeneration.
The Neuro Chip is a two-dimensional (2D) platform. That is, the cells are cultured on microfluidic devices in planar platforms made of small chambers and channels. The cells are kept alive through perfusion of oxygenated and nutrient media. But these 2D platforms with chambers, channels and media do not mimic the natural conditions. The cells do not grow naturally in the same shape and form, do not produce complete networks, and cannot be structured in the actual brain or spinal cord tissue form or in their true functional form. The realistic in vivo platform should have a three-dimensional (3D) structure. Constructing a 3D platform, typically made of porous and hydrogel materials, is an active area of investigation and technological development. Future work in this area will include co-culturing a wide variety of neurons, their accompanying cells such as microglia and Schwann cells, and providing perfusion through microvessels mimicking blood flowing capillaries. Although this field is in its nascence; its future may well lie in NeuroChips built from 3D multi-material scaffold structures, microfluidic channels mimicking capillaries, and multi-cellular neural engineering (http://www.nibib.nih.gov/news-events/newsroom/bioengineers-create-functional-3d-brain-tissue). The NeuroChip technology, extended into 3D, may serve as a stepping-stone for modeling or reverse engineering brain in vitro from its constitutive cellular components.
For Further Reading
1. Y. Xia and G. M.Georgeside, “Soft lithography,” Annual Rev. Materials Sci., Vol. 28: 153-184, 1998.
2. J. W. Park, H. J. Kim, M. W. Kang, N. L. Jeon, “Advances in microfluidics-based experimental methods for neuroscience research. Lab Chip, 13(4):509-21, 2013.
3. I. H. Yang, D. Gary, M. Malone, S. Dria, T. Houdayer, V. Belegu, J. Mcdonald, and N. V. Thakor, “Axon myelination and electrical stimulation in a microfluidic, compartmentalized cell culture platform,” NeuroMolecular Med., 14: 112-118, 2012.
4. S. Hosmane, A. Fournier, R. Wright, L. Rajbhandari, R. Siddique, I. H. Yang, R. K. Ramesh, A. Venkatesan and N. V. Thakor, “Valve-based microfluidic compression platform: single axon injury and regrowth,” Lab Chip, 11 (22): 3888-3895, 2011.
5. E. M. Hur, et al, “Inhibition of nonmuscle myosin II promotes axon growth over inhibitory molecules by regulating growth cone dynamics and microtubule structures ” Proc. Nat. Acad. Sci., 10.1073/pnas.1011258108, 2011.
6. S. Hosmane, I. H. Yang, A. Ruffin, N. Thakor, A. Venkatesan, “Circular compartmentalized microfluidic platform: Study of axon-glia interactions,” Lab Chip, 10(6): 741-747, 2010.
7. R. Siddique, and N. V. Thakor, “Investigation of nerve injury through Microfluidic Devices,” J. Royal Soc. Interface, 11:90, 2014.
Contributor
 Nitish V. Thakor (F’1994) is a Professor of Biomedical Engineering at Johns Hopkins University, Provost Chair Professor at National University of Singapore, and also the Director of the Singapore Institute for Neurotechnology (SINAPSE) at the National University of Singapore. His expertise is in the field of Neurotechnology and Medical Instrumentation. Read more
Nitish V. Thakor (F’1994) is a Professor of Biomedical Engineering at Johns Hopkins University, Provost Chair Professor at National University of Singapore, and also the Director of the Singapore Institute for Neurotechnology (SINAPSE) at the National University of Singapore. His expertise is in the field of Neurotechnology and Medical Instrumentation. Read more







 Nitish V. Thakor (F'1994) is a Professor of Biomedical Engineering at Johns Hopkins University, Provost Chair Professor at National University of Singapore, and also the Director of the Singapore Institute for Neurotechnology (SINAPSE) at the National University of Singapore. His expertise is in the field of Neurotechnology and Medical Instrumentation.
Nitish V. Thakor (F'1994) is a Professor of Biomedical Engineering at Johns Hopkins University, Provost Chair Professor at National University of Singapore, and also the Director of the Singapore Institute for Neurotechnology (SINAPSE) at the National University of Singapore. His expertise is in the field of Neurotechnology and Medical Instrumentation.  Nigel Lovell is Scientia Professor at the Graduate School of Biomedical Engineering, University of New South Wales (UNSW), Sydney, Australia.
Nigel Lovell is Scientia Professor at the Graduate School of Biomedical Engineering, University of New South Wales (UNSW), Sydney, Australia.  José del R. Millán the Defitech Professor at the Ecole Polytechnique Fédérale de Lausanne (EPFL) where he explores the use of brain signals for multimodal interaction and, in particular, the development of non-invasive brain-controlled robots and neuroprostheses. In this multidisciplinary research effort, Dr. Millán is bringing together his pioneering work on the two fields of brain-machine interfaces and adaptive intelligent robotics. He received his Ph.D. in computer science from the Univ. Politècnica de Catalunya (Barcelona, Spain) in 1992.
José del R. Millán the Defitech Professor at the Ecole Polytechnique Fédérale de Lausanne (EPFL) where he explores the use of brain signals for multimodal interaction and, in particular, the development of non-invasive brain-controlled robots and neuroprostheses. In this multidisciplinary research effort, Dr. Millán is bringing together his pioneering work on the two fields of brain-machine interfaces and adaptive intelligent robotics. He received his Ph.D. in computer science from the Univ. Politècnica de Catalunya (Barcelona, Spain) in 1992.  Dr Cuntai Guan received his Ph.D. degree in Electrical and Electronic Engineering from Southeast University in 1993. He is currently Principal Scientist and Department Head at the Institute for Infocomm Research, Agency for Science, Technology and Research (A*STAR), Singapore. His current research interests include neural and biomedical signal processing; neural and cognitive process and its clinical application; brain-computer interface algorithms, systems and its applications.
Dr Cuntai Guan received his Ph.D. degree in Electrical and Electronic Engineering from Southeast University in 1993. He is currently Principal Scientist and Department Head at the Institute for Infocomm Research, Agency for Science, Technology and Research (A*STAR), Singapore. His current research interests include neural and biomedical signal processing; neural and cognitive process and its clinical application; brain-computer interface algorithms, systems and its applications.  Dr Kai Keng Ang received his Ph.D. degree in computer engineering from Nanyang Technological University, Singapore. He is currently the Head of Brain-Computer Interface Laboratory and a Scientist with the Institute for Infocomm Research, Agency for Science, Technology and Research, Singapore. His current research interests include brain-computer interfaces, computational intelligence, machine learning, pattern recognition, and signal processing.
Dr Kai Keng Ang received his Ph.D. degree in computer engineering from Nanyang Technological University, Singapore. He is currently the Head of Brain-Computer Interface Laboratory and a Scientist with the Institute for Infocomm Research, Agency for Science, Technology and Research, Singapore. His current research interests include brain-computer interfaces, computational intelligence, machine learning, pattern recognition, and signal processing.  Mr. Christopher Kuah is a Principal Occupational Therapist currently holding the post of Allied Health Coordinator at the Centre for Advanced Rehabilitation Therapeutics at Tan Tock Seng Hospital. He received his professional qualification in 1995 and attained the Master of Science in Neurorehabilitation from Brunel University (UK) in 2002. His key neurorehabilitation interests include management of clients with hemiplegia and those with complex neuro-disability as a result of stroke and acquired brain injuries. The current focus of his clinical and research work involves development of clinical programs incorporating rehabilitation technologies encompassing robotics, brain-computer interface, and sensor technologies for post-stroke upper limb recovery.
Mr. Christopher Kuah is a Principal Occupational Therapist currently holding the post of Allied Health Coordinator at the Centre for Advanced Rehabilitation Therapeutics at Tan Tock Seng Hospital. He received his professional qualification in 1995 and attained the Master of Science in Neurorehabilitation from Brunel University (UK) in 2002. His key neurorehabilitation interests include management of clients with hemiplegia and those with complex neuro-disability as a result of stroke and acquired brain injuries. The current focus of his clinical and research work involves development of clinical programs incorporating rehabilitation technologies encompassing robotics, brain-computer interface, and sensor technologies for post-stroke upper limb recovery.  Dr. Effie Chew is Senior Consultant in Rehabilitation Medicine, Division of Neurology, University Medicine Cluster, National University Hospital (NUH), Singapore. She received her MBBS from the University of Melbourne, Australia and her Membership to the Royal College of Physicians (Edinburgh, UK). She completed her Advanced Specialist Training in Rehabilitation Medicine in Singapore and went on to complete a Fellowship in Clinical Neurorehabilitation at Spaulding Rehabilitation Hospital, Harvard Medical School, where she worked in the areas of noninvasive brain stimulation for neuromodulation and neurorecovery, as well as neuropharmacology for cognitive recovery in traumatic brain injury and robotics and motor learning in recovery post-stroke. These continue to be her current areas of research interests.
Dr. Effie Chew is Senior Consultant in Rehabilitation Medicine, Division of Neurology, University Medicine Cluster, National University Hospital (NUH), Singapore. She received her MBBS from the University of Melbourne, Australia and her Membership to the Royal College of Physicians (Edinburgh, UK). She completed her Advanced Specialist Training in Rehabilitation Medicine in Singapore and went on to complete a Fellowship in Clinical Neurorehabilitation at Spaulding Rehabilitation Hospital, Harvard Medical School, where she worked in the areas of noninvasive brain stimulation for neuromodulation and neurorecovery, as well as neuropharmacology for cognitive recovery in traumatic brain injury and robotics and motor learning in recovery post-stroke. These continue to be her current areas of research interests.  Dr Karen Sui Geok Chua, MBBS (Singapore), FRCP (Edin), FAMS, is currently senior consultant rehabilitation physician practicing at the Tan Tock Seng Hospital Rehabilitation Centre, Singapore. Her special interests include Neurorehabilitation, Spasticity management, including Botulinum toxin therapy, neurolytic blocks and rehabilitation robotics and technology translation.
Dr Karen Sui Geok Chua, MBBS (Singapore), FRCP (Edin), FAMS, is currently senior consultant rehabilitation physician practicing at the Tan Tock Seng Hospital Rehabilitation Centre, Singapore. Her special interests include Neurorehabilitation, Spasticity management, including Botulinum toxin therapy, neurolytic blocks and rehabilitation robotics and technology translation.