Microchamber for Broadband Electrical Detection of Biological Cells
By Cristiano Palego, Caterina Merla, Yaqing Ning, Caroline Multari, Xuanhong Cheng and James C. M. Hwang
This article presents a novel electrode microchamber with low-loss and matched impedance across the DC-3GHz band. The device incorporates a microfluidic channel that delivers single cells precisely. Prototypes were manufactured using a standard thin film process while maintaining high compatibility with CMOS technology. As proof of concept, live and dead cells were separated through both time-domain and frequency-domain measurements.
New sensing approaches are needed to improve detection of biological hazard, such as pathogens and toxins, as well as for discrimination of normal vs malignant cells. The most widely used biosensing techniques are optical and electrical. While common in laboratories, optical approaches usually require elaborate instruments and cell labeling that can be slow, expensive and cytotoxic. By contrast, electrical biosensors offer the advantage of compactness, robustness and ease of mass production. Most electrical techniques operate by measuring changes in electrical impedance due to the status of the target cells on top of specially designed electrodes. Cells are delivered to the electrodes by either fluidic path [1] or by immersing the electrodes in a biological solution and using bioreceptors that selectively bind to different cells [2]. The delivered cells alter the measured impedance by insulating the electrodes and/or by changing the conductance of the surrounding fluid due to ions leakage. The capacitance and resistance extracted from measured impedance correlate to cytoplasm membrane properties and integrity. Commercially available impedance biosensors often require tens to thousands of cells, whereas applications such as pathogen detection and drug resistance testing require single-cell sensitivity. Assessing the intrinsic cell response without interference from the background solution is another challenge in electrical sensing. To achieve single-cell sensitivity, microfluidic technology that allows reducing the overall volume of suspension around the electrodes is often used in conjunction with narrowband resonance measurement at microwave frequencies. This is also natural because the multi- scale nature of the sensor translates to narrowband impedance matching. However, broadband measurement is more desirable, so that cell external layers can be sensed at low frequencies while subcellular structures can be differentiated at high frequencies. Furthermore, nanosecond electric pulses, which are inherently broadband, may be used for nanoporation and subcellular probing without ionization or heating [3].
This article presents a novel electrode microchamber with low-loss and matched impedance across the DC-3GHz band [4]. The device incorporates a microfluidic channel that delivers single cells precisely. Prototypes were manufactured using a standard thin film process while maintaining high compatibility with CMOS technology. As proof of concept, live and dead Jurkat cells were separated through both time-domain and frequency-domain measurements. This research aims at developing a lab-on-a-chip handheld device that would enable label-free nondestructive biohazard detection at high throughput and without specific biomedical infrastructure or training. Similar point-of-care devices could be someday optimized to allow physicians to detect pathogens from single droplets of bodily fluids. As shown in Fig. 1, the microchamber combines a gold coplanar waveguide fabricated on a quartz microscope slide and a microfluidic channel fabricated in a polydimethylsiloxane (PDMS) cover. In the microchannel region the average spacing between the signal and the ground lines is approximately 15 µm which is comparable to the size of Jurkat cells. The waveguide input and output are terminated in SMA coaxial connectors that can be readily connected to RF measurement equipment while compatible with optical microscopy. To enable cell trapping while decreasing exposure of the waveguide to the fluid, the microchannel crosses the waveguide perpendicularly. Trapping occurs mainly by applying AC signals to the waveguide to induce dielectrophoresis. Additionally, under the microchannel the waveguide has a wavy pattern that helps to enhance trapping mechanically and to concentrate the electric field locally.
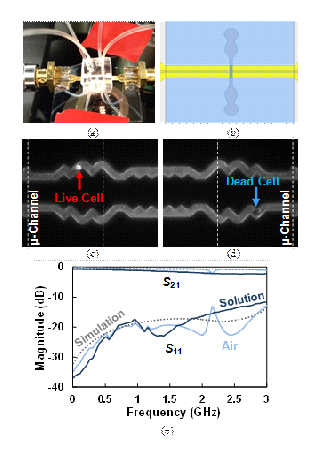
Figure 1: Present microchamber under test. (b) Microfluidic channel with a common inlet cross the coplanar waveguide perpendicularly in the middle. (c) One live cell trapped in a microchannel. (d) One dead cell trapped in a microchannel. (e) Measured (solid curves) and simulated (dotted curves) transmission (S21) and reflection (S11) ratios with the micro-channel empty or filled with a sucrose solution. The little difference in the measured performance with the microchannels empty or filled with an isotonic, ion-free sucrose solution is due to the small overlap between microfluidic channel and electrodes. The simulated curves with empty and filled microchannel are on top of each other.
Fig. 1 confirms that, owing to the small overlap between the microfluidic channel and electrodes, there is little difference in the measured microwave signals with the microchannels empty or filled with an isotonic, ion-free sucrose solution. Additionally, low-loss and impedance matching performance can be achieved up to 3 GHz. With either live or dead Jurkat cells suspended in the sucrose solution, the microchamber impedance was measured between 100 Hz and 100 MHz by using a precision impedance analyzer. The measurements were repeated on the supernatant solutions after filtering out the live or dead cells. Fig. 2 shows that the extracted shunt capacitances and resistances exhibit the well-known dispersion with a flat band between 10 kHz and 1 MHz. Also, the differences between solutions containing live and dead cells consistently exceed the differences between the respective supernatants. The capacitance is consistently higher for live cells, which reflects membrane integrity. However, the extracted shunt resistance is consistently higher for dead cells, which contradicts reported observations [5]. Indeed, in previous researches thousands of cells in a solution are measured simultaneously, hence, as cells die, their membranes disintegrate and ions leak out making the surrounding solution more conductive. In the present case, the ions leaking out of dead cells were washed away before measurement and the measured result reflects the intrinsic properties of cells, whose cytoplasm resistance is higher because fewer ions remain inside the cells.
In addition to quantitative differences in impedance, live and dead cells exhibit qualitative differences in time-domain measurements using ±1.5-V, 20-ns electric pulses with 2-ns rise/fall times and 2% duty-cycle. Bipolar signals were used for simultaneous sensing and trapping without electrode polarization. Fig. 2 shows the incident and reflected signals for solutions with trapped live and dead cells, respectively. With relevant frequency content within 100 MHz, the reflection is mainly due to the shunt capacitance of the live cells with their membranes intact. From the solution with trapped live cells, three reflection peaks associated with rise and fall of the incident bipolar pulse can be clearly delineated. In comparison, from the solution with trapped dead cells, the reflections are buried in background noise. With the broadband microchamber, the incident waveform can be further optimized to better separate live and dead cells.
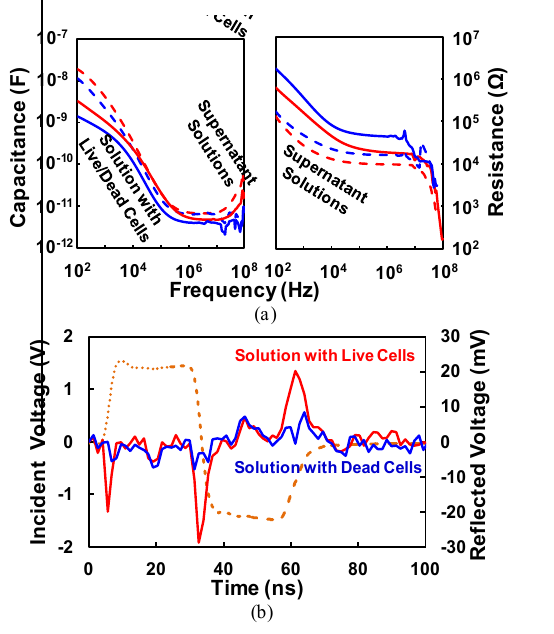
Figure 2: (a) Extracted capacitances and resistances with the microchannels filled with solution with trapped live cells (red solid curve), solution with trapped dead cells (blue solid curve), supernatant solution without live cells (red dotted curve), and supernatant solution without dead cells (blue dotted curve). (b) Incident (dotted curve) and reflected signals from solutions with trapped live (red solid curves) and dead cells (blue solid curves), respectively.
In conclusion, a broadband microchamber incorporating a microfluidic channel was developed for biological cell manipulation and detection with single-cell and label-free operation. The measured capacitance and resistance of live cells were higher and lower, respectively, than those of dead cells, reflecting their membrane integrity and intact ion concentration. This suggests that the biosensor is less hindered by the parasitic solution effect. Broadband time-domain measurement shows interesting potential in separating live and dead cells and proves the capability to couple multiple electrical or even optical, detection techniques.
Acknowledgements
This work was sponsored in part by the Department of the Defense, Defense Threat Reduction Agency, under Grant No. HDTRA1-12-1-0007. The content of the information does not necessarily reflect the position or the policy of the federal government, and no official endorsement should be inferred.
For Further Reading
1. A. U. Hofmann, S. Michaelis, T. Winckler, J. Wegener, and K.-H. Feller, “A whole-cell biosensor as in vitro alternative to skin irritation tests,” Biosensor Bioelectronics, vol. 39, issue 1, pp. 156-62, Jan. 2013.
2. R.L. Caygill, G. E. Blair, P.A. Millner, “A review on viral biosensors to detect human pathogens”, Analytica Chimica Acta, vol. 681, pp. 8-15, November 2010.
3. P. T. Vernier, M. J. Ziegler, Y. Sun, M. A. Gundersen, and D. P. Tieleman, “Nanopore-facilitated, voltage-driven phosphate-idylserine translocation in lipid bilayers – in cells and in silico,” Physical Biology, vol. 3, pp. 233-247, Nov. 2006.
4. C. Palego, C. Merla, Y. Ning, D. Molinero, C. Multari, X. Cheng, G. Ding, and J. C. M. Hwang, “Broadband microchamber for electrical detection of live and dead biological cells,” in IEEE MTT-S Int. Microwave Symp., June 2013.
5. H.-L. Gou, X.-B. Zhang, N. Bao, J.-J. Xu, X.-H. Xia, and H.-Y. Chen, “Label- free electrical discrimination of cells at normal, apoptotic and necrotic status with a microfluidic device,” J. Chromatography. A, vol.1218, pp. 5725-5729, 2011.
Contributors
 Dr. Cristiano Palego is a senior lecturer in Smart Sensors and Instrumentation at Bangor University, UK and a visiting research scientist at Lehigh University, USA. He received the Ph.D. degree in Microwave Engineering and Optoelectronics from the University of Limoges, France. He is currently a research scientist at Lehigh. His interests include electromagnetic theory, micro /nanotechnology and biomedical research as well as RF-MEMS for reconfigurable frontends, antenna arrays, and high-power applications. Read More
Dr. Cristiano Palego is a senior lecturer in Smart Sensors and Instrumentation at Bangor University, UK and a visiting research scientist at Lehigh University, USA. He received the Ph.D. degree in Microwave Engineering and Optoelectronics from the University of Limoges, France. He is currently a research scientist at Lehigh. His interests include electromagnetic theory, micro /nanotechnology and biomedical research as well as RF-MEMS for reconfigurable frontends, antenna arrays, and high-power applications. Read More
 Dr. Caterina Merla received the Ph.D. degrees in electronic engineering from the University of Rome “La Sapienza,” Italy. She is currently with the Italian National Agency for New Technologies, Energy and Sustainable Economic Development (ENEA), Research Centre in Rome and a Visiting Research Scientist at Lehigh University, Bethlehem, PA. Her research interests are mainly focused on the microdosimetric evaluation of the electromagnetic (EM) field at single cell level, biological sample dielectric measurements, and design and dosimetry of exposure systems oriented to EM protection studies and medical applications. Read More
Dr. Caterina Merla received the Ph.D. degrees in electronic engineering from the University of Rome “La Sapienza,” Italy. She is currently with the Italian National Agency for New Technologies, Energy and Sustainable Economic Development (ENEA), Research Centre in Rome and a Visiting Research Scientist at Lehigh University, Bethlehem, PA. Her research interests are mainly focused on the microdosimetric evaluation of the electromagnetic (EM) field at single cell level, biological sample dielectric measurements, and design and dosimetry of exposure systems oriented to EM protection studies and medical applications. Read More
 Yaqing Ning is currently working toward her Ph.D. degree at Lehigh University. She is involved in research on RF MEMS capacitive switches and phase shifters, as well as on biomedical devices for detection purposes. She specializes in 3D electromagnetic simulation using finite element methods. Read More
Yaqing Ning is currently working toward her Ph.D. degree at Lehigh University. She is involved in research on RF MEMS capacitive switches and phase shifters, as well as on biomedical devices for detection purposes. She specializes in 3D electromagnetic simulation using finite element methods. Read More
 Caroline Multari is currently working towards her Ph.D. at Lehigh University in Materials Science and Engineering, focusing on research in point-of-care microfluidic biosensors and developing polymeric biomaterials for bioparticle capture. Read More
Caroline Multari is currently working towards her Ph.D. at Lehigh University in Materials Science and Engineering, focusing on research in point-of-care microfluidic biosensors and developing polymeric biomaterials for bioparticle capture. Read More
 Dr. Xuanhong Cheng is the Rossin Assistant professor of Bioengineering and Materials Science and Engineering at Lehigh University. She received her Ph.D. in bioengineering at University of Washington. She joined Lehigh University, where her current research interest is focused on developing new nanomaterials and microfluidic platforms to analyze intact, live bioparticles, such as cells and pathogens at the point of need. Read More
Dr. Xuanhong Cheng is the Rossin Assistant professor of Bioengineering and Materials Science and Engineering at Lehigh University. She received her Ph.D. in bioengineering at University of Washington. She joined Lehigh University, where her current research interest is focused on developing new nanomaterials and microfluidic platforms to analyze intact, live bioparticles, such as cells and pathogens at the point of need. Read More
 Prof. James C. M. Hwang received the Ph.D. degree in materials science from Cornell University, Ithaca, NY. He joined Lehigh University, Bethlehem, PA as a Professor of electrical engineering and Director of the Compound Semiconductor Technology Laboratory. His current research interests include MEMS, microwave and photonic devices and integrated circuits, and bio-electromagnetics. Read More
Prof. James C. M. Hwang received the Ph.D. degree in materials science from Cornell University, Ithaca, NY. He joined Lehigh University, Bethlehem, PA as a Professor of electrical engineering and Director of the Compound Semiconductor Technology Laboratory. His current research interests include MEMS, microwave and photonic devices and integrated circuits, and bio-electromagnetics. Read More







 J.-C. Chiao is Greene endowed professor and Garrett endowed professor of Electrical Engineering at University of Texas - Arlington; and an Adjunct Associate Professor in the Internal Medicine Department at UT-Southwestern, Medical Center.
J.-C. Chiao is Greene endowed professor and Garrett endowed professor of Electrical Engineering at University of Texas - Arlington; and an Adjunct Associate Professor in the Internal Medicine Department at UT-Southwestern, Medical Center.  Mohammad-Reza Tofighi is an associate professor of electrical engineering with the Capital College, Pennsylvania State University, Middletown, PA. He conducts research on wireless implants, biomedical antennas, medical applications of microwave radiometry, and complex permittivity measurement of tissues using time and frequency domain methods.
Mohammad-Reza Tofighi is an associate professor of electrical engineering with the Capital College, Pennsylvania State University, Middletown, PA. He conducts research on wireless implants, biomedical antennas, medical applications of microwave radiometry, and complex permittivity measurement of tissues using time and frequency domain methods.  Bill Saltzstein is the President of connectBlue Inc., a leading provider of industrial and medical wireless solutions. Bill is also the Medical Business Development Director and joined connectBlue in February of 2011 with a vision to extend medical care out of the traditional environments through the use of wireless technologies.
Bill Saltzstein is the President of connectBlue Inc., a leading provider of industrial and medical wireless solutions. Bill is also the Medical Business Development Director and joined connectBlue in February of 2011 with a vision to extend medical care out of the traditional environments through the use of wireless technologies.  Katia Grenier (S'99 -M'03) received her Ph.D. degree in electrical engineering from the University of Toulouse, France. She is now with the LAAS-CNRS lab, in France, where her research interests are focused on the development of fluidic-based microsystems for biological and medical applications as well as for reconfigurable wireless.
Katia Grenier (S'99 -M'03) received her Ph.D. degree in electrical engineering from the University of Toulouse, France. She is now with the LAAS-CNRS lab, in France, where her research interests are focused on the development of fluidic-based microsystems for biological and medical applications as well as for reconfigurable wireless.  David Dubuc (S'99, M'03) received the Ph.D. degree in electrical engineering from the University of Toulouse, Toulouse, France. He is an Associate Professor with the University of Toulouse, and a Researcher with the Laboratory of Analysis and Architecture of System part of National Scientific Research Center (LAAS-CNRS), Toulouse, France. His research interests include the development of microwave circuits integrated due to microtechnologies and their application to wireless telecommunication and biology.
David Dubuc (S'99, M'03) received the Ph.D. degree in electrical engineering from the University of Toulouse, Toulouse, France. He is an Associate Professor with the University of Toulouse, and a Researcher with the Laboratory of Analysis and Architecture of System part of National Scientific Research Center (LAAS-CNRS), Toulouse, France. His research interests include the development of microwave circuits integrated due to microtechnologies and their application to wireless telecommunication and biology.  Mary Poupot received her PhD degree in Biochemistry from the University of Paul Sabatier, Toulouse, France. She is a Researcher at the Cancer Research Center of Toulouse. Her research interests are based on the impact of the tumor microenvironment on the survey of cancer cell in particular in hematopoietic diseases.
Mary Poupot received her PhD degree in Biochemistry from the University of Paul Sabatier, Toulouse, France. She is a Researcher at the Cancer Research Center of Toulouse. Her research interests are based on the impact of the tumor microenvironment on the survey of cancer cell in particular in hematopoietic diseases.  Jean-Jacques Fournié received his Ph.D. degree in microbial biochemistry from the University of Toulouse, Toulouse, France. He is currently heading the Cancer Research Center of Toulouse (CRCT), France. His fields of scientific expertise are biochemistry, pharmacology, immunology and cancer.
Jean-Jacques Fournié received his Ph.D. degree in microbial biochemistry from the University of Toulouse, Toulouse, France. He is currently heading the Cancer Research Center of Toulouse (CRCT), France. His fields of scientific expertise are biochemistry, pharmacology, immunology and cancer.  Gustavo Guarin is currently pursuing his PhD in Electric Engineering at the University of Erlangen-Nuremberg. He works at the Institute for Electronic Engineering with Prof. Dr.-Ing. Dr.-Ing. habil. Robert Weigel and with Prof. Dr.-Ing. Georg Fischer. The focus of his research is on Ultra-Wideband Microwave Sensors for spectroscopy applications in biomedicine.
Gustavo Guarin is currently pursuing his PhD in Electric Engineering at the University of Erlangen-Nuremberg. He works at the Institute for Electronic Engineering with Prof. Dr.-Ing. Dr.-Ing. habil. Robert Weigel and with Prof. Dr.-Ing. Georg Fischer. The focus of his research is on Ultra-Wideband Microwave Sensors for spectroscopy applications in biomedicine.  Maximilian Hofmann received the Dipl.-Ing. degree in electrical, electronic and communications engineering from the University of Erlangen-Nuremberg, Nuremberg, Erlangen, Germany. He joined the Institute for Electronics Engineering, Erlangen, Germany, as a Research Assistant.
Maximilian Hofmann received the Dipl.-Ing. degree in electrical, electronic and communications engineering from the University of Erlangen-Nuremberg, Nuremberg, Erlangen, Germany. He joined the Institute for Electronics Engineering, Erlangen, Germany, as a Research Assistant.  Dietmar Kissinger received the Dr.-Ing. degree in electrical engineering from the University of Erlangen-Nuremberg, Erlangen, Germany. He holds a position as Lecturer and Head of the RF Integrated Sensors Group at the Institute for Electronics Engineering. His research interests include silicon-based microwave and millimeter-wave integrated circuits as well as wireless sensors and communication systems for ultra-low power, automotive, industrial, security, and medical applications.
Dietmar Kissinger received the Dr.-Ing. degree in electrical engineering from the University of Erlangen-Nuremberg, Erlangen, Germany. He holds a position as Lecturer and Head of the RF Integrated Sensors Group at the Institute for Electronics Engineering. His research interests include silicon-based microwave and millimeter-wave integrated circuits as well as wireless sensors and communication systems for ultra-low power, automotive, industrial, security, and medical applications.  Dr. Cristiano Palego is a senior lecturer in Smart Sensors and Instrumentation at Bangor University, UK and a visiting research scientist at Lehigh University, USA. He received the Ph.D. degree in Microwave Engineering and Optoelectronics from the University of Limoges, France. He is currently a research scientist at Lehigh. His interests include electromagnetic theory, micro/nanotechnology and biomedical research as well as RF-MEMS for reconfigurable frontends, antenna arrays, and high-power applications.
Dr. Cristiano Palego is a senior lecturer in Smart Sensors and Instrumentation at Bangor University, UK and a visiting research scientist at Lehigh University, USA. He received the Ph.D. degree in Microwave Engineering and Optoelectronics from the University of Limoges, France. He is currently a research scientist at Lehigh. His interests include electromagnetic theory, micro/nanotechnology and biomedical research as well as RF-MEMS for reconfigurable frontends, antenna arrays, and high-power applications.  Dr. Caterina Merla received the Ph.D. degrees in electronic engineering from the University of Rome "La Sapienza," Italy. She is currently with the Italian National Agency for New Technologies, Energy and Sustainable Economic Development (ENEA), Research Centre in Rome and a Visiting Research Scientist at Lehigh University, Bethlehem, PA. Her research interests are mainly focused on the microdosimetric evaluation of the electromagnetic (EM) field at single cell level, biological sample dielectric measurements, and design and dosimetry of exposure systems oriented to EM protection studies and medical applications.
Dr. Caterina Merla received the Ph.D. degrees in electronic engineering from the University of Rome "La Sapienza," Italy. She is currently with the Italian National Agency for New Technologies, Energy and Sustainable Economic Development (ENEA), Research Centre in Rome and a Visiting Research Scientist at Lehigh University, Bethlehem, PA. Her research interests are mainly focused on the microdosimetric evaluation of the electromagnetic (EM) field at single cell level, biological sample dielectric measurements, and design and dosimetry of exposure systems oriented to EM protection studies and medical applications.  Yaqing Ning is currently working toward her Ph.D. degree at Lehigh University. She is involved in research on RF MEMS capacitive switches and phase shifters, as well as on biomedical devices for detection purposes. She specializes in 3D electromagnetic simulation using finite element methods.
Yaqing Ning is currently working toward her Ph.D. degree at Lehigh University. She is involved in research on RF MEMS capacitive switches and phase shifters, as well as on biomedical devices for detection purposes. She specializes in 3D electromagnetic simulation using finite element methods.  Caroline Multari is currently working towards her Ph.D. at Lehigh University in Materials Science and Engineering, focusing on research in point-of-care microfluidic biosensors and developing polymeric biomaterials for bioparticle capture.
Caroline Multari is currently working towards her Ph.D. at Lehigh University in Materials Science and Engineering, focusing on research in point-of-care microfluidic biosensors and developing polymeric biomaterials for bioparticle capture.  Dr. Xuanhong Cheng is the Rossin Assistant professor of Bioengineering and Materials Science and Engineering at Lehigh University. She received her Ph.D. in bioengineering at University of Washington. She joined Lehigh University, where her current research interest is focused on developing new nanomaterials and microfluidic platforms to analyze intact, live bioparticles, such as cells and pathogens at the point of need.
Dr. Xuanhong Cheng is the Rossin Assistant professor of Bioengineering and Materials Science and Engineering at Lehigh University. She received her Ph.D. in bioengineering at University of Washington. She joined Lehigh University, where her current research interest is focused on developing new nanomaterials and microfluidic platforms to analyze intact, live bioparticles, such as cells and pathogens at the point of need.  Prof. James C. M. Hwang received the Ph.D. degree in materials science from Cornell University, Ithaca, NY. He joined Lehigh University, Bethlehem, PA as a Professor of electrical engineering and Director of the Compound Semiconductor Technology Laboratory. His current research interests include MEMS, microwave and photonic devices and integrated circuits, and bio-electromagnetics.
Prof. James C. M. Hwang received the Ph.D. degree in materials science from Cornell University, Ithaca, NY. He joined Lehigh University, Bethlehem, PA as a Professor of electrical engineering and Director of the Compound Semiconductor Technology Laboratory. His current research interests include MEMS, microwave and photonic devices and integrated circuits, and bio-electromagnetics.  Marco Mercuri is currently working towards the Ph.D degree within the TELEMIC research group of the Department of Electrical Engineering (ESAT), KU Leuven, Belgium. His research interests include biomedical applications of microwave/RF, wireless sensors, and microwave/millimeter-wave measurements.
Marco Mercuri is currently working towards the Ph.D degree within the TELEMIC research group of the Department of Electrical Engineering (ESAT), KU Leuven, Belgium. His research interests include biomedical applications of microwave/RF, wireless sensors, and microwave/millimeter-wave measurements.  Ping Jack Soh is currently working towards his Ph.D in the ESAT-TELEMIC Research Division, Katholieke Universiteit Leuven, Belgium. His research interests include planar antennas, flexible/textile antennas, on-body communication, metamaterials, passive microwave components and microwave measurements.
Ping Jack Soh is currently working towards his Ph.D in the ESAT-TELEMIC Research Division, Katholieke Universiteit Leuven, Belgium. His research interests include planar antennas, flexible/textile antennas, on-body communication, metamaterials, passive microwave components and microwave measurements.  Dominique Schreurs received the Ph.D. degree in electronic engineering from University of Leuven (KU Leuven), Belgium. She is a Full Professor at KU Leuven. Her main research interests concern the (non)linear characterization and modelling of microwave devices and circuits, as well as (non)linear hybrid and integrated circuit design for telecommunications and biomedical applications.
Dominique Schreurs received the Ph.D. degree in electronic engineering from University of Leuven (KU Leuven), Belgium. She is a Full Professor at KU Leuven. Her main research interests concern the (non)linear characterization and modelling of microwave devices and circuits, as well as (non)linear hybrid and integrated circuit design for telecommunications and biomedical applications.