Barriers to Commercialization of Novel Medical Technologies in India
By Ritu Kamal, Christine Kurihara, Anurag Mairal
A survey of medical device professionals in India reveals the key barriers to increasing access to appropriate medical technologies in the country. We recommend some policy-level changes to facilitate this access through increased commercialization of new medical devices.
The Indian medical device market is valued at about $3 billion, growing 15% per year[1]. It is a relatively small market to serve a population of over 1.3 billion people, reflecting the extremely low utilization of medical technology in the Indian healthcare system. Further, the medical device market in India is dominated by imports, which account for approximately 75% of the devices sold in the country[2] and are priced out of the reach of most patients. This presents an opportunity to medical device innovators who are looking to address the disease burden in India by increasing access to appropriate medical technologies. However, the challenges of the Indian healthcare sector – poor infrastructure, lack of trained personnel, relatively high cost of services, and so on – delay or deny the delivery of suitable medical technology to the Indian patient, particularly by hindering commercialization of new medical products.
In order to examine the barriers to commercialization of novel medical technologies, we surveyed over 200 professionals affiliated with the medical device sector in India to quantitatively determine which of these barriers are most commonly responsible for limiting the development of the market and how they may be mitigated for better commercialization outcomes. The top-three barriers cited by survey respondents were a lack of sufficient early-stage financing, inadequate regulations around manufacture and sale of medical devices, and the lack of intellectual property (IP) protection for new technologies. For instance, as the figure below shows, over 90% of survey respondents consider the lack of funding to be a critical barrier to commercialization. Private interviews with medical technology entrepreneurs and investors looking to enter the Indian market noted the absence of a vibrant ecosystem to support the innovation of medical devices in India. Specific concerns were also raised about the lack of a defined pathway for clinical testing of medical products, a paucity of professionals with knowledge and experience of medical technology, and the fragmentation of distribution networks, which hinders the scaling of medical product sales across the country. Further studies are underway to fully characterize the difficulties faced by companies in different stages of the innovation lifecycle.
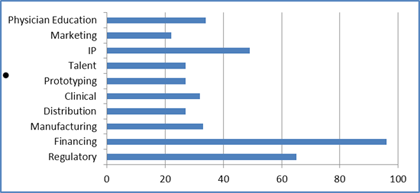
Critical barriers to commercialization as perceived by survey respondents.
We recommend several policy-level changes to incentivize innovators and reduce barriers to commercialization of new medical devices. First, a better regulatory regime is urgently needed; this regime must create a system of control, separate from that of drugs, for quality, safety and efficacy of medical devices in India. Approval of the Medical Devices Regulation Bill [3], already drafted and presented to the Parliament first in 2006 and then again in 2012, will be an important first step in streamlining all laws concerning medical devices in India. Second, adequate IP enforcement, especially in the fragmented segments of the medical technology ecosystem such as prototyping shops and distributors, would protect and encourage innovation in the Indian market. Third, medical technology should be introduced to medical students as part of their coursework at the undergraduate level (MBBS or similar) so that the future doctors are aware of the range of therapies that benefit their patients and are open to adopting new technologies as part of their practice of medicine. Fourth, engineering students should be exposed to medical technology in order to create a pool of skilled engineers who can develop new medical devices appropriate for the Indian setting. Last and perhaps most importantly, different stakeholders of the Indian medical technology industry, such as startup companies, investors, entrepreneurs, multinational companies, management schools, medical and engineering colleges and government organizations, should embrace opportunities to engage with each other and build a collaborative network to help develop and commercialize new medical technologies. The Indian growth story, which is slated to add 500 million people into the middle class by 2025, up from 50 million today[4], presents enormous opportunities for healthcare innovation, and the medical technology industry must seize them.
For Further Reading
1. “India Medical Device Market Intelligence Report 2011,” Espicom Business Intelligence, ISSN 2043-9792
2. “India: Pacific Bridge Medical” http://www.pacificbridgemedical.com/countries/india/
3. “India: New Medical Devices Regulation Bill,” Pacific Bridge Medical, 2011 http://www.pacificbridgemedical.com/news/india-new-medical-devices-regulation-bill-and-foreign-facility-inspections/
4. “Bird of Gold,” McKinsey Institute, 2007







 Nitish V. Thakor is a Professor of Biomedical Engineering at Johns Hopkins University, Baltimore, USA, as well as the Director of the newly formed institute for neurotechnology, SiNAPSE, at the National University of Singapore.
Nitish V. Thakor is a Professor of Biomedical Engineering at Johns Hopkins University, Baltimore, USA, as well as the Director of the newly formed institute for neurotechnology, SiNAPSE, at the National University of Singapore.  Atam Dhawan, Ph.D. obtained his B.Eng. and M. Eng. degrees in Electrical Engineering from the University of Roorkee, Roorkee, India...
Atam Dhawan, Ph.D. obtained his B.Eng. and M. Eng. degrees in Electrical Engineering from the University of Roorkee, Roorkee, India...  Clifford Dacso, MD, MPH, MBA is Professor of Molecular and Cell Biology and Medicine at Baylor College of Medicine, John S. Dunn Sr. Research Chair in General...
Clifford Dacso, MD, MPH, MBA is Professor of Molecular and Cell Biology and Medicine at Baylor College of Medicine, John S. Dunn Sr. Research Chair in General...  Ritu Kamal works at Stanford Biodesign Program, Stanford, CA, as a Global Research Project Manager. She has previously worked at Roche Diagnostics as a Scientist and was a 2010...
Ritu Kamal works at Stanford Biodesign Program, Stanford, CA, as a Global Research Project Manager. She has previously worked at Roche Diagnostics as a Scientist and was a 2010...  Christine Kurihara is Manager of Special Projects, for the Stanford Biodesign Program, Stanford, CA. In this capacity, she oversees IT, web and infrastructure projects for the program.
Christine Kurihara is Manager of Special Projects, for the Stanford Biodesign Program, Stanford, CA. In this capacity, she oversees IT, web and infrastructure projects for the program.  Dr. Anurag Mairal is the Director, Global Exchange Programs, Stanford University, Stanford, CA. In this role, he is responsible for developing an extensive network...
Dr. Anurag Mairal is the Director, Global Exchange Programs, Stanford University, Stanford, CA. In this role, he is responsible for developing an extensive network...  Dr. T. Sunder is a Senior Consultant Cardiothoracic and Transplant Surgeon in Apollo Hospitals, Chennai, India with over 20 years' experience in Cardiothoracic Surgery...
Dr. T. Sunder is a Senior Consultant Cardiothoracic and Transplant Surgeon in Apollo Hospitals, Chennai, India with over 20 years' experience in Cardiothoracic Surgery...