Dynamics and Control for the Artificial Pancreas
Francis J. Doyle III
University of California at Santa Barbara, USA
Reprinted from The Impact of Control Technology
T. Samad and A.M. Annaswamy (eds.), IEEE Control Systems Society, 2011
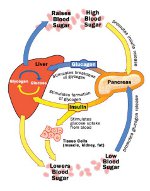
Healthy Regulation of Blood Glucose
The human body uses a combination of opposing manipulated variables (dual control) to achieve regulation of blood sugar, much the same way the driver uses the brake and gas pedals in an automobile. Insulin functions as the “brake pedal,” lowering the blood sugar by stimulating the uptake of glucose from muscle, fat, and kidney cells. Balancing this is the counter-regulatory hormone glucagon, which acts primarily to break down glycogen in the liver, yielding glucose and an elevation in blood sugar levels (acting as the “gas pedal”). Insulin and glucagon are both produced by the pancreas. Although type 1 diabetes is currently incurable, the development of a reliable artificial pancreas would considerably improve the lifestyle of subjects with this disease.
Why Is Systems and Control Relevant?
The systems and control community can play a critical role in developing architectures for the reliable automation of blood glucose monitoring in several ways:
- Advanced control design (for example, model predictive control)
- Design of “verifiable” algorithms for regulatory approval processes
- Safety and fault analysis for medical delivery systems
- Algorithms to monitor the patient and the health of the system
- Advanced glucose calibration algorithms
The Artificial Pancreas Vision
In addition to control algorithms, an automated, fully closed-loop device will require sensors and actuators. Recent developments in continuous blood glucose monitoring (sampling rates of approximately 1—5/min) and innovations in insulin pumps (including telemetry) are promising in this context—the enabling technologies for control engineering to make an impact are well along in development!
Type 1 Diabetes
- Type 1 diabetes is an autoimmune disease leading to insufficient or no production of insulin by the pancreas. The disease causes wide swings in blood glucose levels.
- Current insulin therapies require frequent user intervention (insulin administration and blood glucose measurements). These typically open-loop methods are often ineffective in maintaining blood glucose in the normal range and frequently result in hypoglycemia (low blood sugar) events due to insulin stacking or overdosing.
- Conversely, hyperglycemia (elevated blood glucose) may lead to long-term vascular complications.
- The common insulin administration route is the subcutaneous one via either multiple daily injection (MDI) or continuous subcutaneous insulin infusion (CSII) pump.
- Type 1 diabetes affects 1 million individuals in the U.S. with associated annual medical costs of $15 billion.
Barriers and Challenges
Several technical, policy, and cultural barriers must be addressed before a viable artificial pancreas can be developed
- Performance metrics for closed-loop control are still a subject of discussion.
- Glucose sensor reliability and accuracy remain an issue.
- Delay in insulin action makes systems sluggish.
- Intrasubject variability is a significant challenge (for example, hour-to-hour changes in insulin sensitivity).
- Closed-loop trials face regulatory hurdles.
Several specific engineering challenges must also be resolved:
- Arrays of glucose sensors that are based on a different operating concept than today’s sensors
- Dual-chamber pumps that will allow delivery of both insulin and glucagon
- Accurate predictive patient models that can be customized for the individual
- Communication and interfacing standards for the artificial pancreas
- Well-defined, clinically oriented benchmark scenarios to evaluate control design
- Faster-acting insulin formulations
The Artificial Pancreas System and Preliminary Closed-Loop Trials
Intensive research is under way in all facets of the artificial pancreas. A recent milestone has been the development of the Artificial Pancreas System (APS©) platform at the University of California at Santa Barbara in collaboration with the Sansum Diabetes Research Institute.
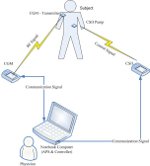
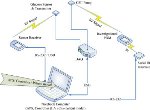
The APS© platform provides a flexible mechanism for integrating hardware (such as glucose sensors and insulin pumps, in addition to computational devices), software, algorithms, and human-machine interfaces.
The APS© is being used in closed-loop trials around the world to test the efficacy of a variety of algorithms for blood glucose control as well as other advanced control applications. It is also being used to link glucose sensors and insulin pumps using wireless protocols (top figure). Combining the APS© with a feedback control algorithm enables both hardware-in-the-loop testing (bottom figure) and closed-loop human clinical trials.