Microfluidics for cancer cell detection and diagnosis
By C.T. Lim
The early development of microfludics in life science applications has been focused on the analysis of biomolecules from minute amount of fluids (typically nanolitres or less). However in recent years, there is increasing use of microfluidics in manipulating and analyzing individual cells, the basic units of life. Here we present an example of a custom microchip that sorts individual Circulating Tumor Cells (CTCs) out of a patient’s blood sample.
The size of individual human cells is typically in tens of microns and microfluidics provides the perfect assay for cell biology and clinical studies. One such study is that of diseased cell detection and analysis. However, some diseased cells are rare, difficult to detect and retrieve, and yet are critically needed for accurate diagnosis. In fact, investigating diseases such as cancer at the single cell level are becoming more and more important as this will enable the detection of minute differences such as gene mutations in individual cancer cells previously not noticeable in analyzing a population of cancer cells. In fact, single cell analysis can potentially contribute towards improved diagnosis, prognosis and treatment. Thus, the need for effective and efficient cell separation has now become an essential first step in many biological and biomedical tests.
Microfluidics has enabled the development of microscale separation techniques in meeting such a need. Several state-of-the-art microfluidic cell separation devices have been developed to detect and diagnose human diseases. There are several important advantages in microfluidics based separation: reduced sample volumes, faster processing time, high sensitivity, low cost and portability. Currently, microfluidic based cell separation devices can be grouped into active or passive devices depending on whether they use an external force field (active) or only microchannel geometry and inherent fluid forces (passive). Active separation techniques include fluorescence-activated cell sorting (FACS), magnetic sorting, dielectrophoresis, electrophoresis, optical and acoustic sorting. Passive approaches involve pillar and weir structures, microfiltration, pinched flow fractionation (PFF), inertial forces and antibody-based affinity.
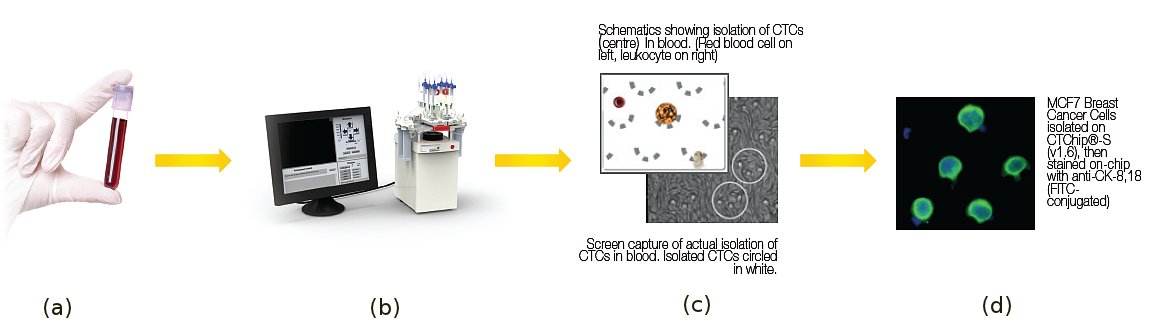
Figure 1: A cancer patient’s blood (a) being processed using the patent-pending microfluidic biochip, CTChip® (b) . Schematic (c) shows how a stiffer and larger CTC is isolated while the more deformable blood cells just squeezed through the cell trap. Image (d) shows stained breast cancer cells trapped in the biochip.

Figure 2: This biochip was developed in our lab and is being commercialized by Clearbridge Biomedics.
We have developed several passive microfluidic cell separation biochips that make use of the fact that CTCs are larger and stiffer than that of blood cells to detect and retrieve CTCs from peripheral blood of cancer patients. Coupled with passive techniques such as microfiltration, PFF and inertial forces, we are able to separate the rare CTCs (typically about 1 to 10 CTCs per billion blood cells) through processing between 2 to 10ml of a cancer patient’s blood (1ml contains ~5 billion blood cells). One example is the CTChip® shown in Figure 2, which we have developed. It makes use of arrays of thousands of crescent-shaped microwells that can isolate CTCs from whole blood without the use of antibodies or magnetic beads. As blood cells flow through the microfluidic chambers containing these microwells, the larger and stiffer CTCs are trapped by the microwells while the smaller and more deformable red and white blood cells simply flows through the arrays of microwells. Once all the blood has been processed, by reversing the microfluidic flow, we are able to dislodge and retrieve the CTCs.
We have developed several passive microfluidic cell separation biochips that make use of the fact that CTCs are larger and stiffer than that of blood cells to detect and retrieve CTCs from peripheral blood of cancer patients. Coupled with passive techniques such as microfiltration, PFF and inertial forces, we are able to separate the rare CTCs (typically about 1 to 10 CTCs per billion blood cells) through processing between 2 to 10ml of a cancer patient’s blood (1ml contains ~5 billion blood cells). One example is the CTChip® shown in Figure 2, which we have developed. It makes use of arrays of thousands of crescent-shaped microwells that can isolate CTCs from whole blood without the use of antibodies or magnetic beads. As blood cells flow through the microfluidic chambers containing these microwells, the larger and stiffer CTCs are trapped by the microwells while the smaller and more deformable red and white blood cells simply flows through the arrays of microwells. Once all the blood has been processed, by reversing the microfluidic flow, we are able to dislodge and retrieve the CTCs.
Clinical tests performed using this biomechanics based biochip have shown it to be more accurate and sensitive in detecting and isolating the extremely heterogeneous CTC population as compared with using the conventional antibody approach which only targets certain subpopulations of CTCs depending on the antibodies used. As every cancer cell counts in the single cell analysis, this will enable researchers and clinicians to have a better understanding of this highly differentiated disease, with vastly different characteristics among patients. A better understanding of these differences will enable physicians to adopt more effective and targeted therapies for cancer management. Finally, obtaining cancer cells from blood (termed liquid biopsy) rather than via the needle aspiration tumor biopsy is extremely attractive as the latter is invasive, painful and cannot be performed on a regular basis.
Microfluidics for such applications is still at its early stage of development but looking ahead, the above example demonstrates how microfluidics can increasingly play an important role in disease detection, diagnosis and prognosis as more of such devices are being translated from bench to bedside.
For Further Reading
H. W. Hou, C. Lee, M. C. Leong, S. Soman, S. R. K. Vedula, C. T. Lim, “Microfluidics for Applications in Cell Mechanics and Mechanobiology”, Cellular and Molecular Bioengineering, Vol. 4, pp. 591-602, 2011.
H. W. Hou, A. A. S. Bhagat, W. C. Lee, S. Huang, J. Y. Han, C. T. Lim, “Microfluidic Devices for Blood Fractionation, Micromachines”, Vol. 2, pp. 319-343, 2011.
A. A. S. Bhagat, H. W. Hou, L. D. Li, C. T. Lim, J. Han, “Pinched flow coupled shear modulated inertial microfluidics with application towards high throughput circulating tumor cell separation”, Lab-on-a-Chip, Vol. 11, pp. 1870-1878, 2011.
C. T. Lim, A. Li, “Mechanopathology of red blood cell diseases – Why mechanics matters”, Theoretical & Applied Mechanics Letters, Vol. 1, Iss. 1, pp. 1-6, 2011.
P. Ang, A. Li, M. Jaiswal, Y. Wang, H. W. Hou, J. Thong, C. T. Lim, K. P. Loh, “Flow Sensing of Single Cell by Graphene Transistor in a Microfluidic Channel”, Nano Letters, Vol. 11, Iss. 12, pp. 5240-6, 2011.
W. C. Lee, A. A. S. Bhagat, H. Sha, K. J. Van Vliet, J. Han, C. T. Lim, “High-throughput cell cycle synchronization using inertial forces in spiral microchannels”, Lab on a Chip, Vol. 11, Iss. 7, pp. 1359 – 1367, 2011.
A. A. S. Bhagat, H. Bow, H. W. Hou, S. J. Tan, J. Y. Han, C. T. Lim, “Microfluidics for cell separation”, Medical & Biological Engineering & Computing, Vol. 48, Iss. 10, pp. 999-1014, 2010.
S. J. Tan, R. L. Lakshmi, P. F. Chen, W. T. Lim, L. Yobas, C. T. Lim, “Versatile label free biochip for the detection of circulating tumor cells from peripheral blood in cancer patients”, Biosensors and Bioelectronics, Vol. 26, pp. 1701 – 1705, 2010.







 In his roles as Chief Medical Officer and Chief Science Officer of the West Wireless Health Institute, Dr. Joseph M. Smith leads initiatives to identify and accelerate the use of health care innovations and technologies...
In his roles as Chief Medical Officer and Chief Science Officer of the West Wireless Health Institute, Dr. Joseph M. Smith leads initiatives to identify and accelerate the use of health care innovations and technologies...  Mathukumalli Vidyasagar received B.S., M.S. and Ph.D. degrees in electrical engineering from the University of Wisconsin in Madison, in 1965, 1967 and 1969 respectively. Between 1969 and 1989, he was a professor of Electrical Engineering at various universities in the USA and Canada.
Mathukumalli Vidyasagar received B.S., M.S. and Ph.D. degrees in electrical engineering from the University of Wisconsin in Madison, in 1965, 1967 and 1969 respectively. Between 1969 and 1989, he was a professor of Electrical Engineering at various universities in the USA and Canada.  C.T. Lim is a Professor of Bioengineering at the National University of Singapore and a Principal Investigator at the university's Mechanobiology Institute.
C.T. Lim is a Professor of Bioengineering at the National University of Singapore and a Principal Investigator at the university's Mechanobiology Institute.  Tom O'Dwyer is Director of Healthcare Technology at Analog Devices Inc. in Wilmington, Massachusetts. In his current role, he has responsibility for all aspects of semiconductor technology for the worldwide healthcare market...
Tom O'Dwyer is Director of Healthcare Technology at Analog Devices Inc. in Wilmington, Massachusetts. In his current role, he has responsibility for all aspects of semiconductor technology for the worldwide healthcare market...