Nanostructured Thin Films Aid Delivery of Promising New Drugs in the Body
By Russell F. Ross and Tejal A. Desai
There is a significant need for new drug delivery approaches to improve the efficacy of drugs. This article highlights the potential for drug delivery systems which use nanostructured thin films to increase the transport of biologics across epithelial tissue and hence improve their distribution and absorption.
Currently, more than 25% of developed drugs never reach commercial status due to poor drug distribution and low absorption levels[1]. These challenges increase immensely for biologics such as proteins, peptides, and siRNA, which are the newest and most promising class of drugs being developed. Biologics have an enormous potential to treat major diseases because they are based on the understanding derived from the human genome. However, they have numerous delivery limitations: their large size reduces diffusion throughout the body and their associated toxicity from higher doses leads to faster clearance, which often prevent them from reaching the intended target tissue. Therefore, to improve the distribution and absorption of biologics, new delivery techniques need to be designed that have more specific control over drug transport mechanisms in epithelial tissue.
Drugs move through epithelial tissue by two primary mechanisms. The first is the paracellular route, which consists of passive diffusion and/or bulk flow around the cells, and the second is the transcellular route, which is transport through the cells using membrane-bound vesicles[2]. The paracellular transport mechanism is governed and limited by the various junctions that seal the epithelial cells together and occlude the interstitial space. The transcellular transport route is an active transport process that begins with drug uptake through vesicles or transporters followed by vesicular sorting and transport through the cell.
It is well known that cells respond to both the topographical and chemical cues invoked by the extracellular matrix (ECM) and neighboring cells. These cues have been shown to affect everything from basic cell function, including shape, migration, adhesion, and survival, to cell proliferation and differentiation[3]. Therefore, it may be possible to perturb epithelial cells with nanostructures to improve the delivery of biological therapeutics by altering the cellular processes related to critical transport mechanisms. The ability to successfully transport high molecular weight proteins across the epithelium by harnessing biological processes has applications in treating numerous diseases currently requiring hypodermic needle injections. This approach to drug delivery is a paradigm shift from the traditional biochemical strategies because it utilizes inert mechanical cues to significantly improve the performance of biological therapies.
In the paper by Kam et al.[4], we show that it is possible to significantly alter the paracellular transport of biologics in vitro, including the 150 KDa biologic Etanercept, by placing nanostructured polymer surfaces in contact with epithelial cells .The results for two different nanostructures are given in Figure 1. These nanostructures differ in their surface roughness, aspect ratio, and elastic modulus. It was found that aspect ratios less than five (5), surface roughness less than 150 nm, and elastic moduli greater than 200 MPa increase permeability and are the key parameters influencing cellular restructuring.
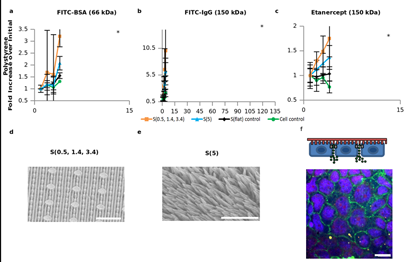
Figure 1: In vitro transport studies. Permeability studies showing how nanostructured surfaces enhance the transport of high molecular weight species across the Caco-2 cell monolayers over 2 hours. Data is displayed as the mean (+/- standard deviation) fold increase over the initial point which is the time when the nanostructured films were placed in contact with the cells. A value of “1” corresponds to no change in drug transport. a, b, and c are the transport of BSA-FITC, IgG-FITC, and etanercept, respectively, across the cell monolayers in contact with the nanostructured polystyrene surfaces. d, nanostructure S(0.5, 1.4, 3.4), e nanostructure s(5), and f fluorescent image of IgG traversing around the cells. Scale bar is 20 µm. Adapted from [4].
The cellular restructuring created by the contact between the nanostructures and epithelial cells can be seen in Figure 2. The mechanical contact modifies the tight junctions that produce a ruffling in the cell wall that increase permeability and drug transport. When the nanostructure surfaced film is removed the cells return to their original morphology demonstrating the reversibility of the biological function.
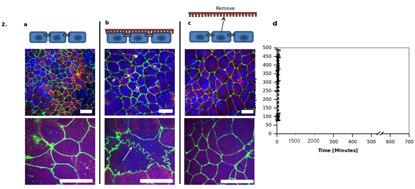
Figure 2: Immunofluorescence staining of the tight junction protein, zonula occluden (ZO-1), was performed. a Staining of the untreated caco-2 monolayer shows a normal cobblestone morphology. b It is apparent that the P(1.5) nanostructured surface induces a dramatic ruffled morphology after 2 hours, indicating tight junction remodeling and a loosening of the epithelial barrier to allow for paracellular transport. c After P(1.5) was removed from the monolayer and incubated for 24 hours, the ZO-1 morphology reverts back to the normal cobblestone architecture, indicating a reversible rearrangement. d TER measurements before and after the nanostructured surface is placed in contact with the cells. TER measurements decreased in the presence of the surface, but eventually increased after 24 hours which demonstrates the reversible and non-deleterious effects of the nanostructures on the cells. Scale bars for the three top images and three bottom images are 10 µm and 20 µm, respectively. Adapted from [4].
In summary, our results suggest that nanoscale topographical cues can be used to increase the permeability of epithelial tissue by placing them in contact with the cells. The nanostructures are influencing drug transport by only mechanical stimulus that actively remodel the tight junction proteins and facilitate the transport of large molecular weight biologics. Without the mechanical stimulus, the biologic cannot be transported in significant quantities across the epithelial cell layer. Furthermore, this phenomenon appears to be a reversible process as indicated by the TEER values which return to their original values when the nanostructured surfaces are removed after 24 hours, suggesting both tight junctions recovery and no cytotoxic effects from the nanostructure. This new transport capability opens up significant new opportunities with particles, implantables, and other drug delivery devices to improve the absorption, distribution, and efficacy of biologics.
For Further Reading
1. “Top 10 Drug Delivery Technologies (2009 – 2014), http://www.marketsandmarkets.com/Market-Reports/top-10-drug-delivery-technologies-112.html
2. Truskey, G. A., F. Yuan, and D.F. Katz, “Transport Phenomena in Biological Systems, 2nd Edition.” Pearson/Prentice Hall, New Jersey, 2009
3. Scheindlin, S., “Transdermal Drug Delivery: PAST, PRESENT, FUTURE.” Molecular Interventions, 4, 308-312 (2004)
4. Kam, K., L. A. Walsh, S. M. Bock, K. E Fischer, M. Koval, R. F. Ross, and T. A. Desai, Nano Lett., November, 2012







 Nitish V. Thakor is a Professor of Biomedical Engineering at Johns Hopkins University, Baltimore, USA, as well as the Director of the newly formed institute for neurotechnology, SiNAPSE, at the National University of Singapore.
Nitish V. Thakor is a Professor of Biomedical Engineering at Johns Hopkins University, Baltimore, USA, as well as the Director of the newly formed institute for neurotechnology, SiNAPSE, at the National University of Singapore.  Ali Khademhosseini, MASc, Ph.D., is an Associate Professor at Harvard-MIT's Division of Health Sciences and Technology (HST), Brigham and Women's Hospital...
Ali Khademhosseini, MASc, Ph.D., is an Associate Professor at Harvard-MIT's Division of Health Sciences and Technology (HST), Brigham and Women's Hospital...  Rashid Bashir he is the Abel Bliss Professor of Electrical and Computer Engineering & Bioengineering, Director of the Micro and NanoTechnology Laboratory...
Rashid Bashir he is the Abel Bliss Professor of Electrical and Computer Engineering & Bioengineering, Director of the Micro and NanoTechnology Laboratory...  Michelle Khine is Associate Professor, Biomedical Engineering; and Associate Professor (Joint Appointment), Chemical Engineering and Materials Science, at the University of California, Irvine.
Michelle Khine is Associate Professor, Biomedical Engineering; and Associate Professor (Joint Appointment), Chemical Engineering and Materials Science, at the University of California, Irvine.  Dr. Russell F. Ross is the Director of Product and Technology Development at Kimberly-Clark Corporation, leading the development of biomaterials for drug delivery and therapeutic applications.
Dr. Russell F. Ross is the Director of Product and Technology Development at Kimberly-Clark Corporation, leading the development of biomaterials for drug delivery and therapeutic applications.  Dr. Tejal Desai is currently Professor of Bioengineering and Therapeutic Sciences at the University of California, San Francisco.
Dr. Tejal Desai is currently Professor of Bioengineering and Therapeutic Sciences at the University of California, San Francisco.  Henry T.K. Tse is a postdoctoral fellow at UCLA where he had also received his Ph.D. in Biomedical Engineering in 2012 under the guidance of Dino Di Carlo.
Henry T.K. Tse is a postdoctoral fellow at UCLA where he had also received his Ph.D. in Biomedical Engineering in 2012 under the guidance of Dino Di Carlo.  Dino Di Carlo is an Associate Professor of Bioengineering at UCLA. He received his B.S. and Ph.D. from UC Berkeley in 2002 and 2006 respectively...
Dino Di Carlo is an Associate Professor of Bioengineering at UCLA. He received his B.S. and Ph.D. from UC Berkeley in 2002 and 2006 respectively...  Daniel Gossett received his Ph.D. from the Biomedical Engineering Interdepartmental Program at the University of California, Los Angeles in the Spring of 2012...
Daniel Gossett received his Ph.D. from the Biomedical Engineering Interdepartmental Program at the University of California, Los Angeles in the Spring of 2012...