Bioengineering at UCSD
By Shankar Subramaniam
NOTE: This is an overview of the entire article, which appeared in the July/August 2012 issue of the IEEE Pulse magazine.
Click here to read the entire article.
Bioengineering is a young albeit important discipline that is still in the process of evolution. Frequently, insightful and prospective students ask two important questions: 1) What is the field of bioengineering and where is it going? 2) Given the diversity of bioengineering and expertise in your department, what is the uniting factor? To answer these, let us ask, what do bio- and biomedical engineers do? Engineers measure components of systems with existing techniques or develop new technologies, understand the design principles of the system, develop a quantitative model, and study the behavior of the system through the model by introducing perturbations. Thus, they are able to build similar systems that can provide similar and different input-response characteristics and lead to innovation. Bioengineers apply these principles to living systems. The end goal is societal and economic benefit, which goes beyond merely satisfying intellectual curiosity.
The article traces the evolution of Bioengineering Program at UCSD from its beginning in 1966 with work on the application of mechanical engineering principles in physiology. Mention is made of the role of the Whitaker Foundation, which supported the building of bio- and biomedical engineering across the country. Tissue engineering has been, and contrinues to be, a strong focus of the program.
Nine current bioengineering research areas at UCSD are highlighted in the article. They are only briefly mentioned in this overview. Refer to the complete article for more information.
(1) First is the work of Dr. Karen Christman and the members of her group, who have been exploring the use of injectable materials to prevent heart failure after a myocardial infarction. Dr. Christman says that a material, termed VentriGel, “appears to prevent further cardiac muscle death, and we saw evidence of stem cells coming into the area of the heart that had been damaged.” This work has led to a commercial company planning clinical trials of the material in Europe.
(2) Dr. Shyni Varghese’s laboratory has been studying stem cell and biomaterials based therapies for treating various musculoskeletal tissue damages. Her laboratory has developed a self-healing hydrogel that binds in seconds, as easily as Velcro, and forms a bond strong enough to withstand repeated stretching. The material has numerous potential applications, including medical sutures, targeted drug delivery, industrial sealants, and self-healing plastics.
(3) Professor Adam Engler and his team have been investigating the in vivo microenvironments that make it difficult to mimic development with functional engineered tissue constructs or engineered “smart cells.” His group explores how these microenvironments affect stem cell niches. Their work with stem cells derived from fat, they suggest, could hint at new therapeutic possibilities for muscular dystrophy (MD).
(4) A 200-patient Phase 2 clinical pilot study was initiated in May 2012 to test the efficacy and safety of a new use and method of administering an enzyme inhibitor for critically ill patients developed by UCSD Department of Bioengineering Professor Geert Schmidt-Schoenbein. Conditions expected to qualify for the study include new-onset sepsis and septic shock, postoperative complications, and new-onset gastrointestinal bleeding. In 2005, the team’s protocol was licensed a development- stage, critical care start-up company.
(5) Dr. Todd Coleman’s lab works on brain-machine interfaces. In cooperation with Dr. John Rogers at the University of Illinois at Champaign/Urbana, they have developed a class of flexible skin-mounted electronics (Figure A) that can be embedded in a temporary tattoo (Figure B). These tattoo sensors that can sense and wirelessly transmit multiple modalities (electrical, temperature, mechanical, and optical).
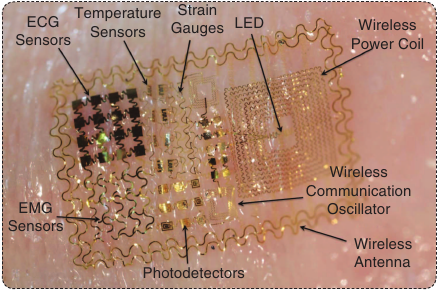
Figure A. The demonstration platform for an epidermal electronics system with multimodal sensing and wireless transmission capability. (Photo courtesy of the University of Illinois.)
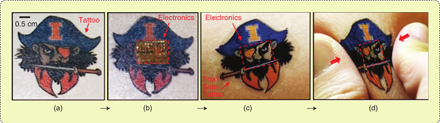
Figure B. Epidermal electronic systems can be embedded in a temporary tattoo that laminates onto and deforms with the skin. (a) Backside of tattoo, (b) after transfer, (c) after integration onto skin, and (d) after deformation. (Photo courtesy of Dr. Todd Coleman, adapted from Science.)
(6) Dr. Bernhard Palsson is a pioneer in microbial systems biology. Palsson and his laboratory have developed genome-scale models of microbes that have revolutionized our understanding of microbial function and are paving the way for a variety of basic and applied research programs from engineering drugs from microbes to making bio-fuels. Palsson’s laboratory built the first complete model of an E. coli organism.
(7) Research from a new bioengineering faculty member, Dr. Christian Metallo, demonstrates that our cells metabolize nutrients in a different manner than has long been thought. “We’re exploring how metabolism controls how our cells function,” said Metallo. “Dysfunctional metabolism is a very important driver of disease, but we often don’t understand the details of what’s going inside the cells and how we can fix the problems that arise when our cells and body are sick. The metabolic pathways we are studying are used by virtually all the cells in our body, so these results actually impact our understanding of metabolism in many different tissues and diseases.”
(8) To better understand how biological clocks are synchronised at the molecular level, Dr. Jeff Hasty created a model biological system consisting of glowing, blinking E. coli bacteria. This simple circadian system allowed Hasty and his team to study in detail how a population of cells synchronizes their biological clocks and enabled the researchers for the first time to describe this process mathematically. To simulate day and night cycles, the researchers modified the bacteria to glow and blink whenever arabinose – a chemical that triggered the oscillatory clock mechanisms of the bacteria – was flushed through the microfluidic chip. This knowledge could have important future applications because scien- tists have discovered that problems with the biological clock can result in many common medical problems ranging from diabetes to sleep disorders.
(9) Dr. Shankar Subramaniam and colleagues have offered the sharpest-yet picture of how core biochemical pathways in skeletal muscle cells and fat cells are altered in people who suffer from insulin resistance – a primary defect in type 2 diabetes and obesity. They have identified major inflammatory and immune regulation pathways that are impaired in insulin resistance that is not addressed by standard treatments. They state that the ability to differentiate between responders and nonresponders to drugs can only be done at a systems level, and this is the quest of quantitative systems pharmacology.
ABOUT THE AUTHOR
Shankar Subramaniam (shankar@ucsd.edu) is the Chair of the Department of Bioengineering, UCSD, and the Joan and Irwin Jacobs Endowed Chair in Bioengineering and Systems Biology.






